Antipsychotic drugs are thought to interfere with sexual functioning but the underlying mechanisms are poorly understood. Those few studies that have been attempted consistently show an association between neuroleptic treatment and sexual dysfunction. The prevalence of sexual dysfunction in groups treated with neuroleptics is thought to be 60% in men and 30-93% in women, with thioridazine being one of the worst culprits (Reference Shader and GrinspoonShader & Grinspoon, 1967; Reference Kotin, Wilbert and VerburgKotin et al, 1976; Reference Ghadirian, Chouinard and AnnableGhadirian et al, 1982; Reference Teusch, Scherbaum and BöhmeTeusch et al, 1995). In addition, sexual dysfunction is worse in patients with schizophrenia taking antipsychotic medication compared with unmedicated patients (Reference Blair and SimpsonBlair & Simpson, 1966; Reference Aizenberg, Zemishlany and Dofman-EhrogAizenberg et al, 1995; Reference Kockott and PfeifferKockott & Pfeiffer, 1996). In this study we attempted to quantify how many patients taking conventional antipsychotic medication complain of sexual dysfunction and to ascertain what mechanisms might be responsible for any sexual dysfunction seen.
METHOD
Assessment
The questionnaires currently available to assess sexual functioning tend to dwell on psychological and relationship aspects of sexual functioning and have not been designed for patients with severe mental illness. Most patients taking antipsychotic medication have schizophrenia, an illness that undermines an individual's ability to relate to other people sexually or otherwise. In this study, we were particularly interested in our subjects' ability to have a physical sexual experience, whether they were in a relationship or not. To this end, a questionnaire was developed — a modified version of that used by Burke et al (Reference Burke, McEvoy and Ritchie1994). Their investigation found that men with schizophrenia were able to answer direct questions regarding concrete aspects of sexual functioning and that there was a significant correlation between score on the questionnaire and measures of dopamine blockade. We modified the questionnaire so that it had sub-scales for the different areas of sexual function and included questions for women. The questionnaire asks about physical aspects of sexual function so that it is not necessary for the subject to have a partner in order to complete it.
The Sexual Functioning Questionnaire (SFQ) that we developed (see Appendix) asks detailed questions about libido, physical arousal (erection in men, vaginal lubrication in women), masturbation, orgasm (including dyspareunia) and ejaculation. It is designed such that higher scores indicate greater dysfunction. It has good reliability: Cronbach's α=0.90; Guttman's split-half reliability=0.86.
As well as sexual function, the following were collected in more detail for the patient group:
-
(a) demographic information;
-
(b) illness history;
-
(c) medication history;
-
(d) depression ratings — using Calgary Depression Inventory (Reference Addington, Addington and Maticka-TyndaleAddington et al, 1992);
-
(e) autonomic side-effects — using UKU questions (Reference Lingaerde, Ahlfors and BechLingaerde et al, 1987);
-
(f) prolactin levels (n=67) — expressed as International Units per litre (IU/1) and measured by chemiluminescent assay on the Bayer Advia ACS:180 (Bayer Diagnostics, Basingstoke).
Subjects
The SFQ was administered to: a consecutive series of out-patient clinic attenders stabilised on antipsychotic medication (n=101); consecutive attenders at a general practice clinic (n=57) who acted as normal controls (mainly presenting with minor respiratory infections); and patients attending a hospital sexual dysfunction clinic (n=55), who acted as ill controls (i.e. individuals with reported and recognised sexual dysfunction). All the clinics are in the same catchment area in South London, served by the Maudsley and King's College Hospitals.
Of the patients who were approached, 8 refused to enter the study and 36 were excluded because of the reasons cited below.
Subjects between the ages of 18 and 60 years stabilised on antipsychotic medication were eligible for entry into the study. Exclusion criteria included diabetes, hypertension, cardiovascular disease, gonadal injury, endocrine disorder/medication, alcohol misuse, antidepressant or anti-convulsant medication, inability to give consent or answer questions and female patients who were peri— or post-menopausal.
Analyses
Results were analysed using SPSS version 7.5 for Windows. The results for males and females were analysed separately because the design of the questionnaire is such that there are different questions and thus different overall scores, depending on gender.
Demographic characteristics and outcome on the SFQ were evaluated for differences across the three groups using one-way analysis of variance (ANOVA) for continuous variables (with 95% CI for difference in means) and chi-squared analysis for categorical variables. Relationships between different variables were assessed with Pearson's correlation coefficient and partial correlations. Analysis of covariance (ANCOVA) was used to ascertain the effect of different variables on the SFQ score. The Bonferroni test was used for post hoc analysis of difference. To correct for the large number of statistical tests conducted in the univariate analysis, the 1% level was used to look for significant effects rather than the 5% level.
RESULTS
Sexual dysfunction scores
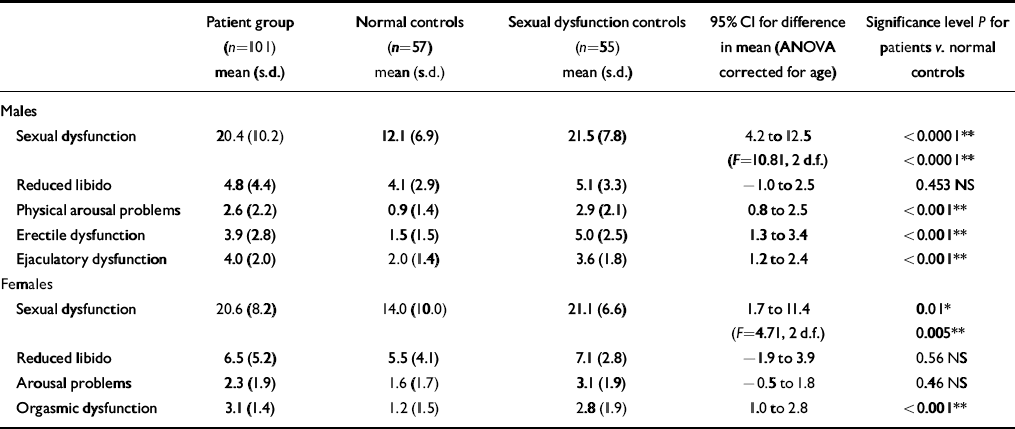
The mean score on the SFQ was 20.5 for patients, 12.6 for normal controls and 21.5 for controls from the sexual dysfunction clinic (the 95% CI for difference in means between patients and normal controls was 4.7-11.1). Table 1 shows the scores in each area of sexual function by group.
Table 1 Scores on the Sexual Functioning Questionnaire by group
| Patient group (n=101) mean (s.d.) | Normal controls (n=57) mean (s.d.) | Sexual dysfunction controls (n=55) mean (s.d.) | 95% CI for difference in mean (ANOVA corrected for age) | Significance level Pfor patients v. normal controls | |
|---|---|---|---|---|---|
| Males | |||||
| Sexual dysfunction | 20.4 (10.2) | 12.1 (6.9) | 21.5 (7.8) | 4.2 to 12.5 (F=10.81, 2 d.f.) | <0.0001** |
| <0.0001** | |||||
| Reduced libido | 4.8 (4.4) | 4.1 (2.9) | 5.1 (3.3) | -1.0 to 2.5 | 0.453 NS |
| Physical arousal problems | 2.6 (2.2) | 0.9 (1.4) | 2.9 (2.1) | 0.8 to 2.5 | <0.001** |
| Erectile dysfunction | 3.9 (2.8) | 1.5 (1.5) | 5.0 (2.5) | 1.3 to 3.4 | <0.001** |
| Ejaculatory dysfunction | 4.0 (2.0) | 2.0 (1.4) | 3.6 (1.8) | 1.2 to 2.4 | <0.001** |
| Females | |||||
| Sexual dysfunction | 20.6 (8.2) | 14.0 (10.0) | 21.1 (6.6) | 1.7 to 11.4 (F=4.71, 2 d.f.) | 0.01* |
| 0.005** | |||||
| Reduced libido | 6.5 (5.2) | 5.5 (4.1) | 7.1 (2.8) | -1.9 to 3.9 | 0.56 NS |
| Arousal problems | 2.3 (1.9) | 1.6 (1.7) | 3.1 (1.9) | -0.5 to 1.8 | 0.46 NS |
| Orgasmic dysfunction | 3.1 (1.4) | 1.2 (1.5) | 2.8 (1.9) | 1.0 to 2.8 | <0.001** |
An arbitrary cut-off point of 1 standard deviation above the mean was taken as the threshold above which sexual dysfunction was said to be present. Using this threshold for normal controls, 45% of the subject group reported sexual dysfunction compared with 17% of normal controls and 61% of controls from the sexual dysfunction clinic.
Patients taking antipsychotics reported levels of libido that did not differ significantly from that of normal controls. Using the criterion stated above for sexual dysfunction, the male patients were 6.3 times as likely to complain of sexual dysfunction as the normal control males (95% CI 1.8-23.4), 3.7 times as likely to complain of erectile dysfunction (95% CI 1.4-10.1) and 16.4 times as likely to complain of ejaculatory dysfunction (95% CI 2.1-128.5). Female patients were 9.6 times more likely to complain of orgasmic dysfunction (95% CI 2.9-32.1) than the normal control females (95% CI 1.3-10.1). There were no significant differences between the level of sexual dysfunction reported by the patient group compared with those attending the sexual dysfunction clinic, although the patient group tended to have better levels of libido. It is of note that the male patients described difficulty in achieving erections, including morning erections and complete inability to achieve erection enough for penetration. Also, the male patients particularly complained of delayed ejaculation or reduced ejaculatory volume. Subjects were asked about masturbatory activity. Male patients reported as much masturbatory activity as normal controls (odds ratio=0.8 (0.7-1.1), NS) and controls from the sexual dysfunction clinic (0.8 (0.7-0.9), NS). Female patients also reported similar levels of masturbatory activity compared with normal controls (odds ratio=0.7 (0.5-1.1), NS) and controls from the sexual dysfunction clinic (0.6 (0.3-1.5), NS).
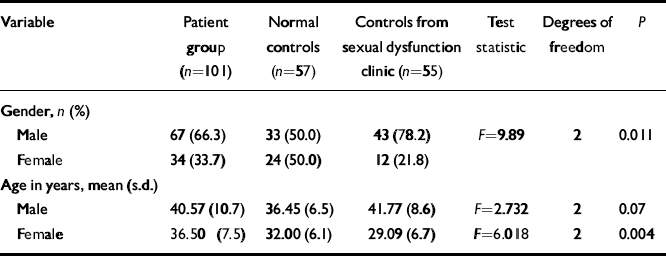
Age and sexual function
Age distributions of the three groups differed in both men and women, although this did not reach significance for men (Table 2). Age and sexual functioning are related such that as age increases, sexual function may worsen. Controlling for age, however, made little difference to the result for males; patients taking antipsychotic medication and controls attending the sexual dysfunction clinic still scored significantly higher on the SFQ. For females, controlling for age actually increased the significance of the F statistic, indicating the effect of a relatively young sample attending the sexual dysfunction clinic (Table 2).
Table 2 Gender and age distribution
| Variable | Patient group (n=101) | Normal controls (n=57) | Controls from sexual dysfunction clinic (n=55) | Test statistic | Degrees of freedom | P |
|---|---|---|---|---|---|---|
| Gender, n(%) | ||||||
| Male | 67 (66.3) | 33 (50.0) | 43 (78.2) | F=9.89 | 2 | 0.011 |
| Female | 34 (33.7) | 24 (50.0) | 12 (21.8) | |||
| Age in years, mean (s.d.) | ||||||
| Male | 40.57 (10.7) | 36.45 (6.5) | 41.77 (8.6) | F=2.732 | 2 | 0.07 |
| Female | 36.50 (7.5) | 32.00 (6.1) | 29.09 (6.7) | F=6.018 | 2 | 0.004 |
Type of medication and sexual function
The different types of neuroleptic medications taken by the patients included depot flupentixol, fluphenazine, haloperidol and zuclopenthixol and oral haloperidol, tri-fluoperazine, thioridazine, sulpiride, chlor-promazine, pimozide and droperidol. The mean dose of medication in chlorpromazine equivalents was 220.10 mg (s.d.=228.33) for males and 280.60 mg (s.d.=290.38) for females. There were no significant differences in mean dose of medication for each group of neuroleptics (ANOVA: F=0.126, P=0.972).
Figures 1 and 2 show a breakdown of the scores on the SFQ for each medication subtype in each area of sexual functioning for men and women. There were no significant differences between medication type and overall mean SFQ score, although there was a tendency (in both men and women) for those taking butyrophenones to report less sexual dysfunction than those taking other drugs. When sexual function was broken down into its components, it can be seen that medication type did not distinguish between male subjects with sexual dysfunction. However, for females, the substituted benzamides and thioxanthenes were more likely to be associated with physical arousal problems than other drugs and the aliphatic phenothiazines and thioxanthenes were more likely to cause anorgasmia than other drugs. This finding was strengthened by controlling for dose of medication.
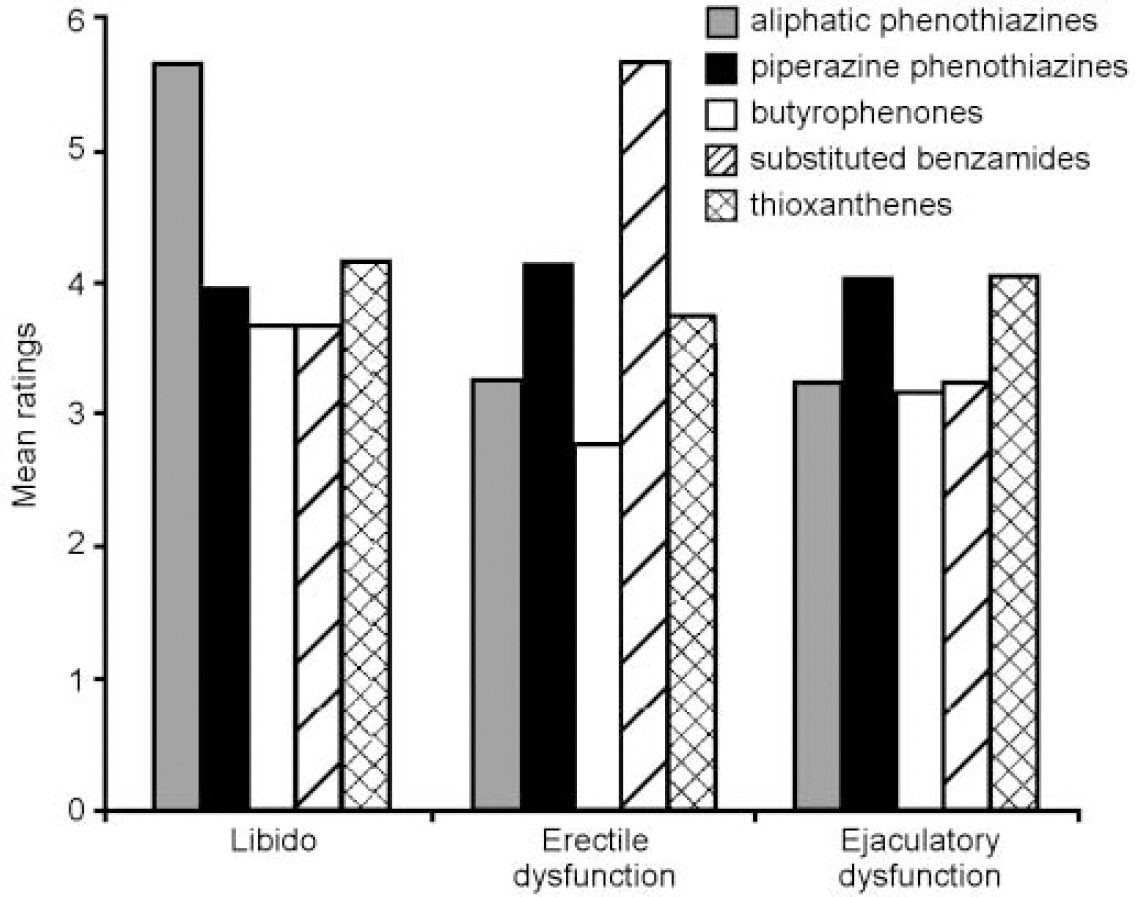
Fig. 1 Sexual dysfunction by type of medication for males.
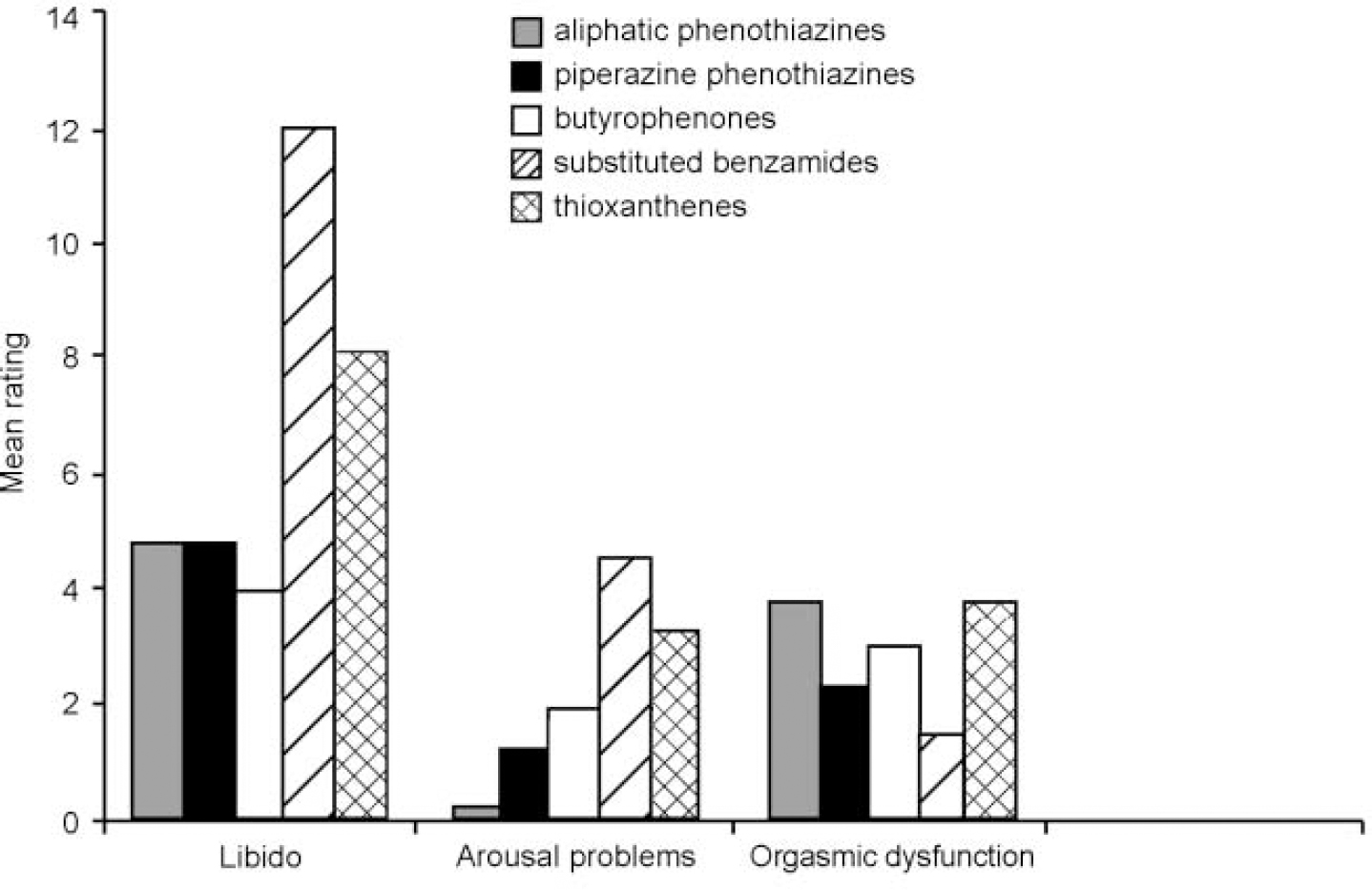
Fig. 2 Sexual dysfunction by type of medication for females: arousal problems, significant group effect (F=4.3, d.f.=4, P=0.009); orgasmic dysfunction, significant group effect (F=2.76, d.f.=4,P=0.049).
Factors associated with sexual dysfunction
Table 3 shows mean depression scores, mean autonomic side-effect scores, mean dose of medication (in chlorpromazine equivalents) and mean prolactin levels for the patient group. Patients had mild to moderate levels of depression and moderate to high rates of autonomic side-effects. For the male patients, prolactin levels were in the high normal range, with 34% being hyperprolactinaemic (prolactin > 480 IU/1). Female patients had mean prolactin levels that were outside the normal range, with 75% being hyperprolactinaemic (prolactin > 480 IU/1).
Table 3 Mean prolactin levels, depression scores, autonomic side-effect scores and medication doses for the patient group

| Males | Females | Significance level P | |
|---|---|---|---|
| Prolactin, IU/I (n=67) | 404 | 1061 | <0.001 |
| CDI (n=101) | 5.4 (s.d.=4.0) | 3.9 (s.d.=3.8) | NS |
| Autonomic side-effects (n=101) | 6.9 (s.d.=5.3) | 6.2 (s.d.=4.8) | NS |
| Medication, mg (n=101) | 221 (222) | 280 (290) | NS |
Correlations between different areas of sexual function and depression score, anticholinergic and antiadrenergic side-effects, dose of medication and prolactin levels showed that, for males, age was significantly correlated with erectile dysfunction (r=0.46, P < 0.001) and ejaculatory dysfunction (r=0.24, P=0.043) but not poor libido, implying that sexual interest does not diminish with age. Depression correlated significantly with poor libido (r=0.37, P=0.007), erectile dysfunction (r=0.34, P=0.0017) and orgasmic dysfunction (r=0.35, P=0.012) but there was no association between depression and ejaculatory function. Dose of medication was significantly correlated with erectile dysfunction (r=0.3, P=0.02). Autonomic side-effects were significantly correlated with male sexual dysfunction, such that anticholineric side-effects were particularly associated with erectile dysfunction (r=0.32, P=0.041) and antiadrenergic side-effects were associated with abnormal ejaculation (retarded ejaculation or reduced volume) (r=0.43, P=0.006). Controlling for dose of medication resulted in these relationships being strengthened (r=0.43, P=0.006 for erectile dysfunction; r=0.53, P < 0.001 for ejaculatory function). For male patients there was no correlation between prolactin and any measure of sexual dysfunction.
Thirty-four per cent of the males were hyperprolactinaemic. For this group, the overall reported sexual dysfunction did not differ from the normoprolactinaemic male patients. However, when sexual function was broken down into its component areas, age, depression and autonomic side-effects ceased to be related to measures of sexual dysfunction, whereas prolactin was negatively associated with erectile dysfunction (r=-0.562, P=0.023) and quality of orgasm (r=-0.56, P=0.023).
For female patients there was no correlation between age and sexual dysfunction or autonomic side-effects and sexual dysfunction. Dose of medication was associated with reduced physical arousal (r=0.36, P=0.05). Depression was significantly correlated with poor libido (r=0.41, P=0.03), reduced physical arousal (r=0.59, P=0.001) and orgasmic problems (r=0.648, P < 0.001). Prolactin correlated negatively with libido (r=-0.46, P=0.03) and physical arousal problems such as poor vaginal response (r=0.52, P=0.02). Controlling for dose of medication reduced the significance of the association between prolactin and libido (r=-0.46, P=0.06), as did controlling for depression (r=-0.37, P=0.17). However, the association between prolactin and poor vaginal response was strengthened by controlling for dose of medication (r=0.6, P=0.011) and for depression (r=0.62, P=0.014).
DISCUSSION
Methodology
Our questionnaire did not address aspects of relationships and it may be that it is not complete to discuss sexual function outside the context of a relationship. However, the majority of patients in this study did not have partners (88.5% never married, separated or divorced, with 71.3% not married or co-habiting). This compares with a British population figure of 29.1% for a similarly aged group who are without partners (Reference WisniewskiOffice for National Statistics, 1999). A questionnaire that included questions about relationships with other people would have resulted in the majority of the patients being unable to enter the study.
The best way of finding out whether antipsychotic medication has a negative effect on sexual function is to compare subjects before and after they start medication. We found that many patients who were drug-free were too unwell to complete a questionnaire asking intimate details about sexual function. The design of this present study still allows for us to estimate the level of sexual dysfunction and also allows us to tease out the mechanisms that might be underlying any sexual dysfunction seen in patients taking antipsychotic medication.
Although there were sufficient numbers to give 99% power to distinguish between the groups in terms of sexual function, the study was only partly able to ascertain the effect of different neuroleptics on the individual areas of sexual function because of the small numbers of patients taking certain drug types.
Sexual function
Patients taking antipsychotics reported high levels of sexual dysfunction, similar to the levels seen in a sexual dysfunction clinic, yet these patients were no more likely to have abnormal sexual interest or to masturbate than normal controls. This appears to counter the commonly held view that psychotic illness ipso facto leads to abnormal sexual interest. However, our finding of normal libido may be in keeping with Aizenberg et al's (Reference Aizenberg, Zemishlany and Dofman-Ehrog1995) view that anti-psychotic medication has a positive effect on psychological functioning and thus allows patients with schizophrenia to have normal sexual interest.
Mechanisms underlying sexual dysfunction in patients taking antipsychotic drugs
For men, we found that increasing age, higher levels of depression, higher doses of medication or worse anticholinergic or antiadrenergic side-effects were all associated with higher levels of reported sexual dysfunction. These relationships ceased to exist when men were hyperprolactinaemic. This indicates that treatment-emergent sexual dysfunction is most commonly associated with the autonomic side-effects of antipsychotic drugs, but, if a man becomes hyperprolactinaemic following antipsychotic treatment, this raised prolactin is likely to be the main cause of any sexual dysfunction seen.
The majority of women in our study were hyperprolactinaemic, they complained of high rates of sexual dysfunction and this was significantly related to their levels of depression, dose of medication or their prolactin level but not to other factors such as age or autonomic side-effects. When depression and dose of medication were controlled, the relationship between prolactin and sexual dysfunction in females strengthened, implying that hyperprolactinaemia is the main cause of sexual dysfunction in females taking antipsychotic medication.
For men, we were unable to differentiate between medications in terms of sexual dysfunction. In women, substituted benzamides (e.g. sulpiride) and thioxanthenes (e.g. flupentixol decanoate) were more likely to be associated with physical arousal problems than other medications, whereas aliphatic phenothiazines (e.g. chlorpromazine) and thioxanthenes were more likely to be associated with anorgasmia.
Sulpiride is a highly selective dopamine blocker; flupentixol, in common with other thioxanthenes, is a potent inhibitor at the dopamine receptor and has few anticholinergic or antiadrenergic effects. Difficulty with physical arousal (e.g. poor vaginal lubrication, as seen in postmenopausal women) is thought to be a result of hypooestrogenaemia and is a common finding in patients with hyperprolactinaemia (Reference BachmannBachmann, 1995; Reference SchlechteSchlechte, 1995). The fact that the women taking substituted benzamides and thioxanthenes were suffering the sexual side-effects commonly seen with hyperprolactinaemia reinforces our finding that this is the main cause of sexual dysfunction in the women taking antipsychotic medication in this study.
Sexual dysfunction and compliance
Non-adherence to antipsychotic medication is one of the main causes of relapse in patients with psychotic illness. Side-effects of medication are frequently cited as a reason for non-compliance. Clinicians generally feel that the worse side-effects of antipsychotic medication are the extra-pyramidal side-effects and those most likely to trouble patients and make it difficult for the person to adhere to their medication regime (Reference Smith and HendersonSmith & Henderson, 2000). Despite this, Finn et al (Reference Finn, Bailey and Schultz1990) showed that patients are more concerned with the sexual side-effects of their medications than any other side-effect. The National Schizophrenia Fellowship have published a survey of over 2000 mental health service users (http://www.nsf.org.uk/information/research). They found that a large proportion of the people had experienced side-effects from their antipsychotic medication and that sexual side-effects were deemed to be the most troublesome.
Clearly, sexual side-effects of medication may be an important cause of non-adherence. The findings from this study give an indication of how this troublesome side-effect may be combated.
CLINICAL IMPLICATIONS
-
• Sexual dysfunction is common in patients taking conventional neuroleptic medication and discussion of sexual side-effects may prevent non-compliance in patients too embarrassed to discuss spontaneously their sexual problems with their doctor.
-
• Clinicians should routinely enquire about sexual symptoms prior to prescription of antipsychotics and on follow-up.
-
• Antipsychotics with fewer effects on prolactin may be less likely to cause sexual dysfunction, especially in women, and therefore these drugs should be used preferentially to avoid sexual difficulties.
LIMITATIONS
-
• We did not include unmedicated patients in the study.
-
• Larger numbers of patients would be required to ascertain the effect of specific medications on sexual function.
-
• Penile or vaginal plethysmography and seminal fluid analysis would have given greater information about organic sexual dysfunction.
APPENDIX
Sexual Functioning Questionnaire

| Pt. No. _____ Date. _____ | |
| Each statement is followed by a TRUE or FALSE answer. Read each statement carefully and decide which response best describes how you feel. Then put a circle round the corresponding response. If you are not completely sure which response is more accurate, circle the response that you feel is most appropriate. Please ask the person interviewing you if there are any words you do not understand. Do not spend too long on each statement. It is important that you answer each question as honestly as possible. Remember to answer every question. | |
| ALL INFORMATION WILL BE TREATED WITH THE STRICTEST CONFIDENCE. | |
| Over the past month | |
| 1. I have thought about sex | |
| a. at least once per day | TRUE/FALSE |
| b. three times per week | TRUE/FALSE |
| c. less than once per week | TRUE/FALSE |
| d. less than once a fortnight | TRUE/FALSE |
| 2. I never think about sex | TRUE/FALSE |
| 3. I have found other people sexually desirable | TRUE/FALSE |
| 4. I have not wanted to have sexual intercourse | TRUE/FALSE |
| 5. I have enjoyed sex | TRUE/FALSE |
| 6. I have not been particularly interested in sex | TRUE/FALSE |
| Over the past month | |
| 7. I have been easily aroused sexually | TRUE/FALSE |
| 8. It has taken longer than usual for me to become sexually aroused | TRUE/FALSE |
| 9. I have been completely unable to become sexually aroused | TRUE/FALSE |
| 10. Although I have been aroused mentally, nothing has happened physically | TRUE/FALSE |
| Please answer this next section only if you are male. | |
| Over the past month | |
| 11. I have had erections (morning erections or erections on awaking) | |
| a. every day | TRUE/FALSE |
| b. about three times per week | TRUE/FALSE |
| c. less than once per week | TRUE/FALSE |
| d. less than once a fortnight | TRUE/FALSE |
| e. less than once per month | TRUE/FALSE |
| 12. I do not have erections | TRUE/FALSE |
| 13. I am always able to achieve a full erection if I want to | TRUE/FALSE |
| 14. I feel that my erections are not as full now as they used to be | TRUE/FALSE |
| 15. I am never able to achieve a full erection | TRUE/FALSE |
| 16. I rarely achieve a full erection | TRUE/FALSE |
| 17. Because I cannot achieve a full erection, I am unable to have intercourse | TRUE/FALSE |
| Please answer this next section only if you are female | |
| Over the past month | |
| 18. Sex has been difficult or painful for me because I do not respond physically as I ought to | TRUE/FALSE |
| 19. My physical response to sexual stimulation is different now to what it used to be | TRUE/FALSE |
| 20. My physical response to sexual stimulation is better now than it used to be | TRUE/FALSE |
| 21. My physical response to sexual stimulation is worse now than it used to be | TRUE/FALSE |
| Over the past month | |
| 22. I have masturbated | |
| a. at least once a day | TRUE/FALSE |
| b. about three times per week | TRUE/FALSE |
| c. about once per week | TRUE/FALSE |
| d. less than once per fortnight | TRUE/FALSE |
| e. less than once per month | TRUE/FALSE |
| 23. I feel that masturbation is wrong | TRUE/FALSE |
| 24. I never masturbate | TRUE/FALSE |
| 25. I rarely masturbate | TRUE/FALSE |
| 26. I have masturbated more often than I usually do | TRUE/FALSE |
| 27. I have masturbated less than I usually do | TRUE/FALSE |
| Over the past month | |
| 28. I have not achieved orgasm/ejaculation by any means at all | TRUE/FALSE |
| 29. I have had orgasms/ejaculations as often as I have wanted | TRUE/FALSE |
| 30. I have never had an orgasm/ejaculation | TRUE/FALSE |
| 31. Orgasm/ejaculation has been painful for me | TRUE/FALSE |
| 32. My orgasm/ejaculation has been different to before | TRUE/FALSE |
| 33. I have an orgasm/ejaculate every time I have sex/masturbate | TRUE/FALSE |
| Please answer the following if you are male | |
| 34. I ejaculate a long time after I have achieved orgasm | TRUE/FALSE |
| 35. My ejaculation happens too quickly | TRUE/FALSE |
| 36. The amount of fluid that I produce when I ejaculate is less than I used to produce before | TRUE/FALSE |
| 37. The amount of fluid that I produce when I ejaculate is more than I used to produce before | TRUE/FALSE |
| 38. The colour of the fluid that I produce when I ejaculate is different to before | TRUE/FALSE |
| Instructions for Interviewer | |
| A. Although this has the appearance of a structured questionnaire, the nature of the topic often means that you will need to clarify terms in order to ensure that the subjects know what is being asked of them. Also, if you are able to talk about sex, the subjects may feel more comfortable when filling in the questionnaire. | |
| B. Remind the subjects that the questions are quite personal, but also normalise the experience by reminding them of the usual process of sexual intercourse and the problems that people might encounter if their sexual function is poor (e.g. “Usually in order for people to have sex, they have to have an erection, some people find that they have difficulties with this. Section three asks questions about this area” or “Some people complain of difficulties with their sex life, they may have problems getting aroused sexually or they can't have orgasms, this questionnaire asks about this kind of thing”). | |
| C. Section Two: sexual arousal involves the mental phenomena of being sexually interested and is usually accompanied by penile erection in males and vaginal lubrication and swelling of the vaginal walls in females (usual physical response after sexual stimulation in women). | |
| D. Terms | |
| Erection — when the penis gets hard or stiff. | |
| Vaginal lubrication — when the vagina becomes moist. | |
| Orgasm — the feeling that happens at the end of sex. This is usually accompanied by overwhelming physical sensations in women, along with vaginal wall contractions. In men it is accompanied by ejaculation. | |
| Ejaculation — the production of seminal fluid or semen. | |
Acknowledgements
Penny Mostyn and Simon Vearnals helped to collect data from patients at the sexual dysfunction clinic. We thank the Myatt's Field Health Centre, London, for their cooperation during the course of this study.










eLetters
No eLetters have been published for this article.