The increasing use of psychotropic medication to treat child and adolescent disorders is causing concern in the community. This particularly applies to the use of stimulants such as amphetamines and methylphenidate. Is the prescription of stimulants restricted to conditions for which they are an effective treatment? Is the medical profession placing too much emphasis on the use of medication to treat childhood mental health problems and failing to utilise effective non-drug treatments? Are worries about overuse of stimulants the reaction of an ill-informed public?
PHARMACO-EPIDEMIOLOGY OF STIMULANTS
Reliable information on the pharmaco-epidemiology of stimulants is scarce, particularly in relation to Europe. The overall prevalence of stimulant use in children has been estimated as 1-6% in the USA (Reference Jensen, Kettle and RoperJensen et al, 1999; Reference Angold, Erkanli and EggerAngold et al, 2000; Reference Barbaresi, Katusic and ColliganBarbaresi et al, 2002), 2% in Australia (Reference Sawyer, Rey and GraetzSawyer et al, 2002), 5% in Canada (Reference PoulinPoulin, 2001) and 0.03% in Britain (Reference KewleyKewley, 1998). There is good evidence that treatment with stimulants has been increasing in the USA, Canada and Australia, although there are large variations between countries and between regions within countries (Reference Jensen, Kettle and RoperJensen et al, 1999).
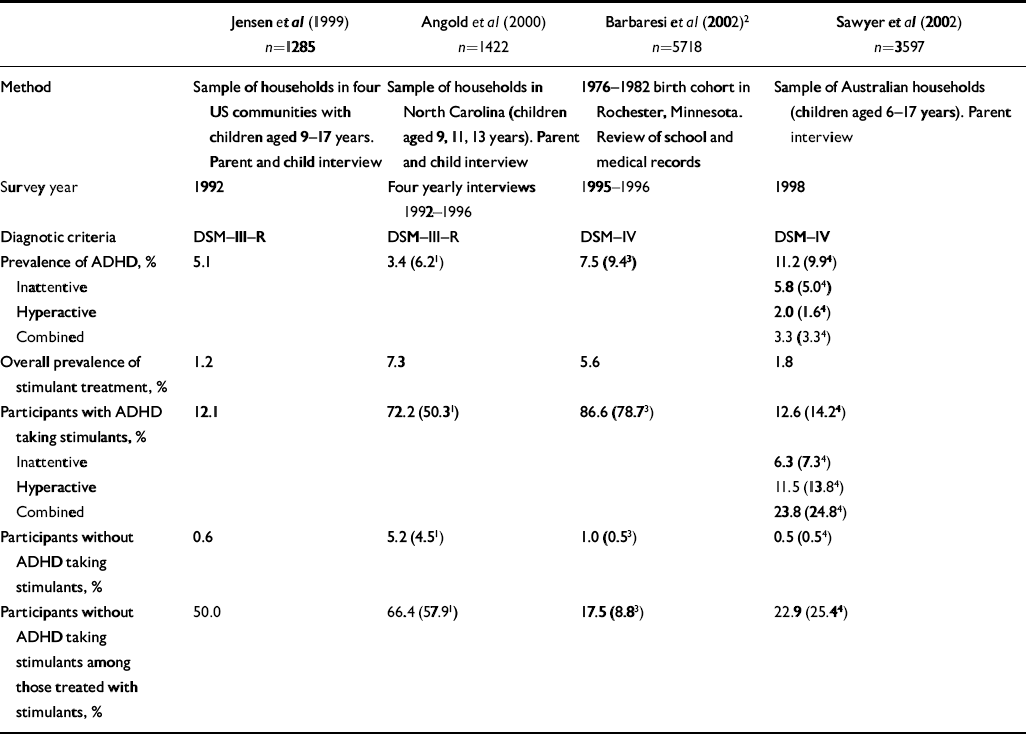
The surveys that have examined community samples of young people with attention-deficit hyperactivity disorder (ADHD) and medication use are summarised in Table 1. Even in countries with higher prescription rates, such as the USA and Australia, many children with ADHD do not receive medication.
Table 1 Prevalence of stimulant treatment and attention-deficit hyperactivity disorder (ADHD) in community surveys
| Jensen et al (Reference Jensen, Kettle and Roper1999) n=1285 | Angold et al (Reference Angold, Erkanli and Egger2000) n=1422 | Barbaresi et al (Reference Barbaresi, Katusic and Colligan2002)2 n=5718 | Sawyer et al (Reference Sawyer, Rey and Graetz2002) n=3597 | |
|---|---|---|---|---|
| Method | Sample of households in four US communities with children aged 9-17 years. Parent and child interview | Sample of households in North Carolina (children aged 9, 11, 13 years). Parent and child interview | 1976-1982 birth cohort in Rochester, Minnesota. Review of school and medical records | Sample of Australian households (children aged 6-17 years). Parent interview |
| Survey year | 1992 | Four yearly interviews 1992-1996 | 1995-1996 | 1998 |
| Diagnotic criteria | DSM-III-R | DSM-III-R | DSM-IV | DSM-IV |
| Prevalence of ADHD, % | 5.1 | 3.4 (6.21) | 7.5 (9.43) | 11.2 (9.94) |
| Inattentive | 5.8 (5.04) | |||
| Hyperactive | 2.0 (1.64) | |||
| Combined | 3.3 (3.34) | |||
| Overall prevalence of stimulant treatment, % | 1.2 | 7.3 | 5.6 | 1.8 |
| Participants with ADHD taking stimulants, % | 12.1 | 72.2 (50.31) | 86.6 (78.73) | 12.6 (14.24) |
| Inattentive | 6.3 (7.34) | |||
| Hyperactive | 11.5 (13.84) | |||
| Combined | 23.8 (24.84) | |||
| Participants without ADHD taking stimulants, % | 0.6 | 5.2 (4.51) | 1.0 (0.53) | 0.5 (0.54) |
| Participants without ADHD taking stimulants among those treated with stimulants, % | 50.0 | 66.4 (57.91) | 17.5 (8.83) | 22.9 (25.44) |
Children without ADHD treated with stimulants
The surveys listed in Table 1 also show that a substantial number of children who did not meet criteria for ADHD were treated with stimulants. Although the population percentages were small (0.5-4%), they represented between 18% and 72% of all young people being prescribed these drugs. Comparison between the studies is not straightforward because diagnostic criteria and methodology varied. Surveys that used DSM—III—R criteria (American Psychiatric Association, 1987) reported prevalences of ADHD of 3.4% and 5.1%. The Australian study (Reference Sawyer, Rey and GraetzSawyer et al, 2002), using DSM—IV criteria (American Psychiatric Association, 1994), showed a rate of 11.2%, or 9.9% if impairment was considered. This rate was similar to that reported by Barbaresi et al (Reference Barbaresi, Katusic and Colligan2002) in the Rochester cohort: a cumulative incidence by age 19 years of 7.5% (or 9.4% if ‘definite’ and ‘probable’ ADHD were included).
Not surprisingly, the diagnostic criteria used have a large impact on the estimates of the number of children with and without ADHD who are treated. For example, if instead of any subtype of ADHD only the combined subtype was used to identify those with ADHD in the Australian study (this information is not available for the other reports), the proportion of children not meeting criteria for ADHD but receiving stimulants would increase from 23% of all those receiving stimulants (14 879 children aged 6-17 years Australia-wide) to 56% (35 032). The combined subtype requires the presence of symptoms of inattention, impulsivity and overactivity. The two other subtypes are new diagnoses introduced in DSM—IV: the inattentive form needs symptoms of inattention only, and the impulsive—hyperactive subtype requires symptoms of impulsivity and overactivity only. The combined subtype is similar (but not identical) to ADHD in DSM—III—R and to hyperkinetic disorder in ICD—10 (World Health Organization, 1992), corresponds to the traditional construct of ADHD, and is the better researched and validated form of the condition (almost all treatment studies involve the participation of patients with this syndrome). Therefore, the most appropriate rate to consider when comparing these studies may be that observed when only the combined subtype is included. The results thus obtained are similar across the studies for which data are available: half or more of all the children treated with stimulants did not meet criteria for the combined subtype (Reference Jensen, Kettle and RoperJensen et al, 1999; Reference Angold, Erkanli and EggerAngold et al, 2000; Reference Sawyer, Rey and GraetzSawyer et al, 2002). That is, although a large proportion of children with ADHD are not being treated with stimulants, up to half of those treated do not have the combined form. A further half of the remainder of those treated probably meet criteria for the inattentive and impulsive—hyperactive subtypes, if the Australian data are to be believed. Some may conclude from this that the number of young people being treated with stimulants in the USA and Australia and who do not have ADHD is unacceptably high.
These conclusions may be incorrect, because instruments used in epidemiological studies may underdiagnose or misdiagnose ADHD, or because a full diagnostic assessment similar to that conducted in clinical practice (in which other information may be considered) is not possible. Further, there are several reasons why children without ADHD may be prescribed stimulants. First, it is possible that they have subsyndromal forms of the condition that nevertheless cause impairment. Second, the troublesome behaviour of some children may be misdiagnosed (e.g. by mistaking non-compliance, learning or conduct problems for ADHD). Third, clinicians in ordinary practice may prescribe stimulant drugs not only for ADHD but also for other emotional and behavioural problems in an attempt to manage difficult children or to enhance school performance. Finally — and most importantly — it may be that stimulant treatment had reduced the symptoms experienced by children with ADHD; hence, they would not meet criteria for diagnosis at the time of interview in epidemiological studies.
Regrettably, there is limited evidence to support or disprove these explanations, and more research is required. However, a multiple-wave study showed that 56% of the treated children without ADHD began receiving medication after the first interview (Reference Angold, Erkanli and EggerAngold et al, 2000). These children had not met criteria for ADHD prior to stimulant treatment. At the assessment before that in which they began stimulant treatment, 71% had no parent-reported ADHD symptoms, 27% had just one and 2% had three. Their counts of ADHD symptoms the following year, while taking stimulant medication, were as follows: 56% had no symptoms, 2% had one, 2% had two, 38% had three and 2% had eleven. The authors concluded that low symptom counts in those started on stimulants during the course of the study could not be accounted for by the curative effect of the drug.
CLINICAL PRACTICE GUIDELINES
Overprescription, underprescription and inappropriate prescription of stimulants co-exist and variations appear to be specific to country, region and provider (Reference Jensen, Kettle and RoperJensen et al, 1999). Stimulants are effective in the short to medium term in reducing the core symptoms of ADHD, as shown by the Multimodal Treatment Study of Children with ADHD (MTA) and many other studies (Reference TaylorTaylor, 1999). Nevertheless, there is debate about the extent to which management of children diagnosed with ADHD should include stimulants, and about the role of psychological interventions (Reference TaylorTaylor, 1999).
The National Institute for Clinical Excellence (NICE) recommends that methylphenidate should be used for treatment of children with severe hyperkinetic disorder (broadly equivalent to the combined form of ADHD). This implies that stimulants should not be recommended for children with the inattentive and impulsive—hyperactive subtypes. The guidance estimates that approximately 1% of children in the community suffer from this severe form of the illness, but NICE gives no further guidelines for their identification (National Institute for Clinical Excellence, 2000). Statements of this kind leave the door open for variations in interpretation. This may be more of a problem when clinicians are not skilled in psychiatric diagnosis, particularly in a condition such as ADHD that relies on reports from informants (e.g. parents) rather than on direct observation or examination of children by medical practitioners.
In contrast, the guidelines of the American Academy of Pediatrics state that once diagnosis is made (any subtype), clinicians should recommend stimulant medication and/or behaviour therapy (American Academy of Pediatrics, 2001). If this were followed, up to 17% of all boys aged 6-12 years might warrant stimulant treatment, according to the Australian survey. Although most clinicians would find this rate unpalatable it is not farfetched: a study in two school districts in Virginia reported that 17% of White boys at primary school were taking stimulant medication (Reference LeFever, Dawson and MorrowLeFever et al, 1999).
CONCLUSIONS
Given the limited data available, particularly from the UK, no general conclusions can be made. Although there is much evidence for the effectiveness of stimulants for the combined subtype of ADHD, concern may be justified about the number of children without ADHD who are being prescribed stimulants in the USA and Australia — mainly because ADHD is a chronic condition and treatment usually lasts years not weeks. Guidelines for the treatment of ADHD are multiplying but they are inconsistent and leave too much scope for interpretation. It is important to discuss where we should draw the line for stimulant treatment: all those who meet criteria for severe hyperkinetic disorder? At any form of ADHD? Should those who ‘almost’ meet criteria be treated also? European countries, where rates of stimulant prescription are lower, can learn from the American and Australian experiences. A first step would be to monitor prescription trends.
Improving accuracy and effectiveness in the treatment of ADHD as well as achieving the right balance between pharmacological and psychological interventions may be a ‘litmus test’. If the medical profession, the pharmaceutical industry and policy-makers fail in this, the backlash — which has already started — may hinder progress in this area, and children will be the losers.






eLetters
No eLetters have been published for this article.