Depression is the most common psychiatric disorder and the fourth major cause of disease burden worldwide (World Health Organization, 2001). In the UK, one in four women and one in ten men experience at least one episode of depression requiring treatment during their lifetime (National Depression Campaign, 1999) and more than half of these will have at least one more episode (Reference KupferKupfer, 1991). Treatment options for moderate and severe depression include pharmacotherapy, psychological therapy and the combination of the two. Since these strategies have different resource requirements and their availability may vary significantly, it is important to evaluate not only their benefits and risks but also their cost-effectiveness. This paper presents an updated version of a decision analysis developed for the National Institute for Clinical Excellence (2004) depression guideline to evaluate the outcomes and likely costs of the first-line use of antidepressant medication or combination therapy for moderate and severe depression in secondary care in the UK (National Collaborating Centre for Mental Health, 2005).
METHOD
There has been little research on the comparative cost-effectiveness of antidepressant therapy and combination therapy. We could identify in the international literature only one study, a trial-based economic evaluation, but this assessed cost-effectiveness of combination therapy for relapse prevention for patients with residual depression (Reference Scott, Palmer and PaykelScott et al, 2003). No estimate exists, however, for the likely outcomes and costs of the routine use of different treatment strategies for patients with moderate and severe depression in secondary care. Therefore, a pragmatic decision analytic model was developed to compare the effectiveness and costs of using antidepressant therapy alone with using combination therapy for people with moderate and severe depression over a 15-month period (3-month initial treatment and 12-month follow-up, no maintenance therapy) (Fig. 1). These time horizons were chosen to be most similar to the available comparative clinical evidence. All analyses were carried out in Microsoft Excel (2003 release).
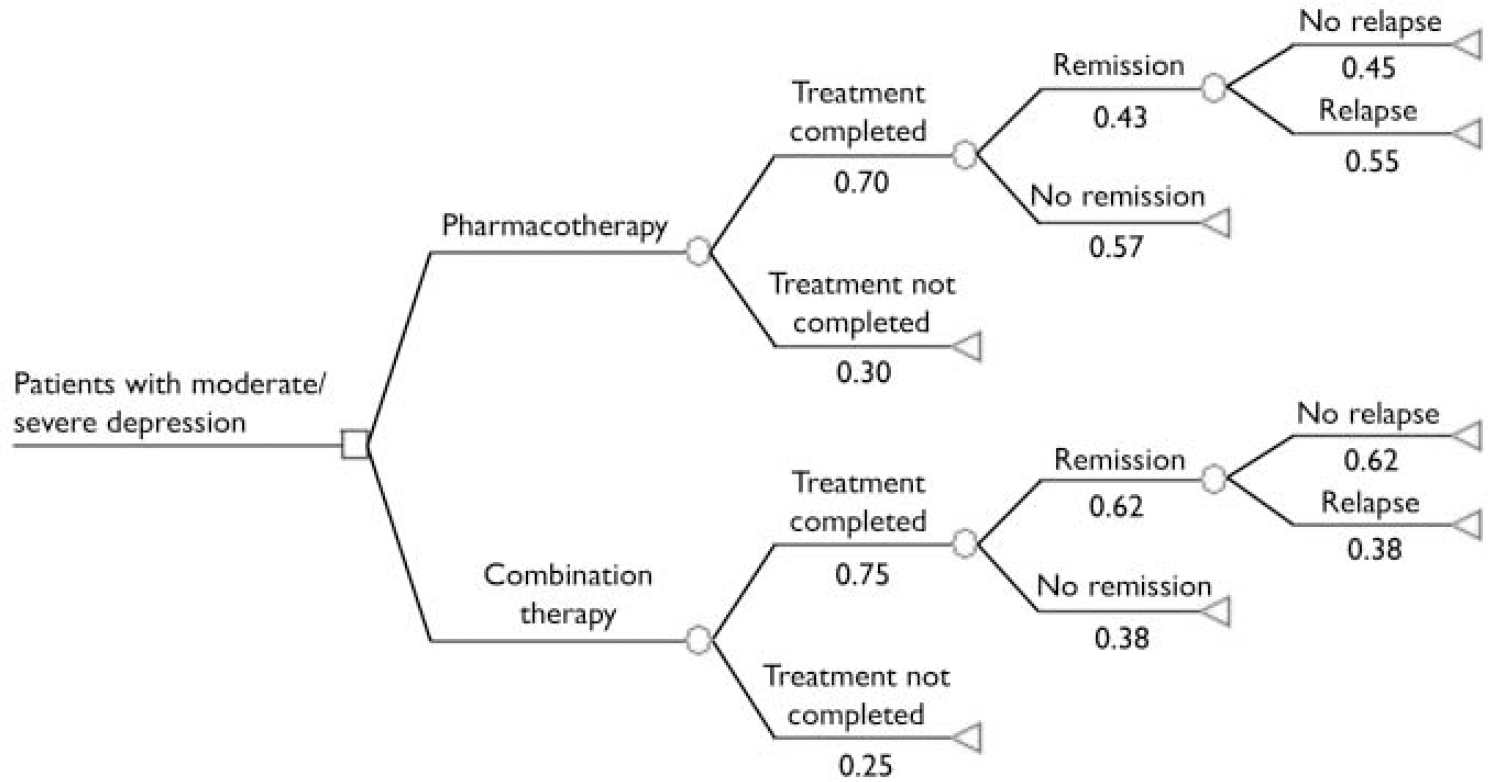
Fig. 1 Decision analysis structure and event probabilities (estimated from the clinical effectiveness parameters listed in Table 1).
Table 1 Decision analysis parameters
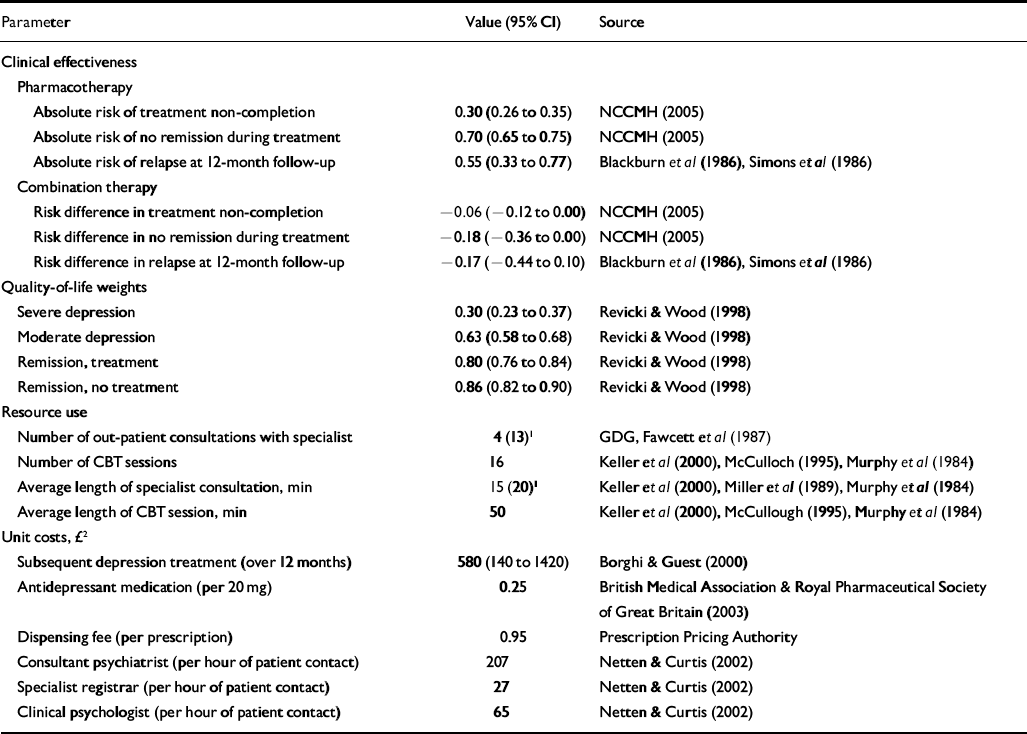
| Parameter | Value (95% CI) | Source |
|---|---|---|
| Clinical effectiveness | ||
| Pharmacotherapy | ||
| Absolute risk of treatment non-completion | 0.30 (0.26 to 0.35) | NCCMH (2005) |
| Absolute risk of no remission during treatment | 0.70 (0.65 to 0.75) | NCCMH (2005) |
| Absolute risk of relapse at 12-month follow-up | 0.55 (0.33 to 0.77) | Blackburn et al (Reference Blackburn, Euson and Bishop1986), Simons et al (Reference Simons, Murphy and Levine1986) |
| Combination therapy | ||
| Risk difference in treatment non-completion | -0.06 (-0.12 to 0.00) | NCCMH (2005) |
| Risk difference in no remission during treatment | -0.18 (-0.36 to 0.00) | NCCMH (2005) |
| Risk difference in relapse at 12-month follow-up | -0.17 (-0.44 to 0.10) | Blackburn et al (Reference Blackburn, Euson and Bishop1986), Simons et al (Reference Simons, Murphy and Levine1986) |
| Quality-of-life weights | ||
| Severe depression | 0.30 (0.23 to 0.37) | Revicki & Wood (Reference Revicki and Wood1998) |
| Moderate depression | 0.63 (0.58 to 0.68) | Revicki & Wood (Reference Revicki and Wood1998) |
| Remission, treatment | 0.80 (0.76 to 0.84) | Revicki & Wood (Reference Revicki and Wood1998) |
| Remission, no treatment | 0.86 (0.82 to 0.90) | Revicki & Wood (Reference Revicki and Wood1998) |
| Resource use | ||
| Number of out-patient consultations with specialist | 4 (13)1 | GDG, Fawcett et al (Reference Fawcett, Epstein and Fiester1987) |
| Number of CBT sessions | 16 | Keller et al (Reference Keller, McCullough and Klein2000), McCulloch (1995), Murphy et al (Reference Murphy, Simons and Wetzel1984) |
| Average length of specialist consultation, min | 15 (20)1 | Keller et al (Reference Keller, McCullough and Klein2000), Miller et al (Reference Miller, Norman and Keinter1989), Murphy et al (Reference Murphy, Simons and Wetzel1984) |
| Average length of CBT session, min | 50 | Keller et al (Reference Keller, McCullough and Klein2000), McCullough (Reference McCullough1995), Murphy et al (Reference Murphy, Simons and Wetzel1984) |
| Unit costs, £2 | ||
| Subsequent depression treatment (over 12 months) | 580 (140 to 1420) | Borghi & Guest (Reference Borghi and Guest2000) |
| Antidepressant medication (per 20 mg) | 0.25 | British Medical Association & Royal Pharmaceutical Society of Great Britain (2003) |
| Dispensing fee (per prescription) | 0.95 | Prescription Pricing Authority |
| Consultant psychiatrist (per hour of patient contact) | 207 | Netten & Curtis (Reference Netten and Curtis2002) |
| Specialist registrar (per hour of patient contact) | 27 | Netten & Curtis (Reference Netten and Curtis2002) |
| Clinical psychologist (per hour of patient contact) | 65 | Netten & Curtis (Reference Netten and Curtis2002) |
Clinical effectiveness data were obtained from a wider review of psychological therapies for the treatment of depression undertaken to support the development of the National Institute for Clinical Excellence (NICE) clinical guideline (National Collaborating Centre for Mental Health, 2005). This recommends cognitive–behavioural therapy and interpersonal therapy as psychological therapies in the treatment of moderate and severe depression. We chose cognitive–behavioural therapy for our analysis since it has a relatively large, high-quality evidence base compared with other psychological therapies provided by the National Health Service (NHS). It is also more widely available than interpersonal therapy, for which there are a number of high-quality trials (National Collaborating Centre for Mental Health, 2005). Originally we also considered a strategy of using cognitive–behavioural therapy alone as first-line treatment. The clinical effectiveness evidence synthesised in the guideline systematic review, however, suggested that this option had no additional clinical advantage in reducing depression symptoms by the end of treatment compared with antidepressants for this patient group. Furthermore, there is insufficient evidence to determine whether there is a clinically significant difference between cognitive–behavioural therapy and antidepressants on reducing the likelihood of relapse (National Collaborating Centre for Mental Health, 2005). As the former therapy has substantially higher initial treatment costs (see below) with no expected savings in the subsequent healthcare resource use compared with antidepressant therapy alone, this strategy was excluded from further evaluation (expert opinion of the Guideline Development Group; see Acknowledgements).
Review of clinical effectiveness
Randomised controlled trials comparing psychological therapies with other active or inactive treatments (either in combination or alone) in the treatment of people with a primary diagnosis of major depressive disorder diagnosed according to DSM or ICD criteria (or equivalent) were identified through systematic literature searches of electronic databases (CINAHL, EM-BASE, Medline, PsycINFO) from inception to January 2002 (updated August 2004) and by using reference lists of existing reviews (further information available from the authors upon request). In addition, reference lists of identified studies were hand-searched and known researchers in the field were contacted for details of unpublished studies. Searches identified 5292 citations. Data for cognitive–behavioural therapy (individual and group), behaviour therapy, short-term psychodynamic psychotherapy, interpersonal therapy, couples therapy, problem-solving therapy and non-directive counselling were reviewed. Trials were included provided the method of treatment allocation was adequate (i.e. the randomisation method used was unbiased so that each participant stood an equal chance of being allocated to any group and the allocated group could not be known before allocation took place) and efficacy ratings were undertaken by a masked assessor. Trials in which some participants had a primary diagnosis of bipolar disorder or dysthymia rather than a primary diagnosis of (unipolar) major depressive disorder were included provided the proportion with the alternative diagnosis did not exceed 15% for bipolar disorder or 20% for dysthymia (figures arrived at through consultation with the Guideline Development Group). In addition, study medication must have been given at the standard dosage or above: either that stated by Bollini et al (Reference Bollini, Pampallona and Tibaldi1999) or, for drugs not included in that meta-analysis, the dosage advised in the British National Formulary (BNF; British Medical Association & Royal Pharmaceutical Society of Great Britain, 2003). Severity of depression symptoms was based on the baseline Hamilton Rating Scale for Depression (HRSD; Reference HamiltonHamilton, 1960) scores. Because of the range of cut-off scores suggested in the literature, we used those proposed by the American Psychiatric Association: no depression 0–7; mild symptoms 8–13; moderate symptoms 14–18; severe symptoms 19–22; very severe symptoms ≥23 (National Collaborating Centre for Mental Health, 2005). Studies were initially assessed by two reviewers working independently (R. B. and Preethi Premkumar or Lisa Underwood (see Acknowledgements)), with a clinical review by expert members of the Guideline Development Group (S. P., Paul Gilbert and Ian Hughes (see Acknowledgements)).
For the current analysis, clinical evidence on the effectiveness of individual cognitive–behavioural therapy in combination with antidepressants compared with antidepressant therapy alone in patients with moderate and severe depression was synthesised using meta-analysis. Efficacy data were extracted and entered into Review Manager software version 4.2.3 (Cochrane Collaboration) by one reviewer (R.B.) and checked by another (Preethi Premkumar or Lisa Underwood). Relapse data used in this analysis were extracted by J.S. Data were not extracted if more than 50% of a treatment group left treatment for whatever reason, to preserve the robustness of the estimated effect sizes. Data were extracted on an intention-to-treat basis so that participants who did not complete the study protocol were treated as having the least favourable outcome. Data were pooled using a fixed-effects model (Reference Hedges and OlkinHedges & Olkin, 1985), unless significant heterogeneity was present (defined as I 2 >50%) that could not be explained by sensitivity analyses (for example, based on differences in study methodology), when a random effects model was used (Reference DerSimonian and LairdDerSimonian & Laird, 1986; Reference Higgins and ThompsonHiggins & Thompson, 2002). Risk differences were calculated for the various effectiveness measures together with their 95% confidence intervals.
Outcomes
The number of patients treated successfully (remission and no relapse over a 12-month follow-up period) was chosen as the primary outcome measure for the decision analysis. Remission was defined as reaching scores 6 or less on the 17-item HRSD or 8 or less on the 24-item HRSD during treatment. Patients with unsuccessful treatment outcomes included those who did not complete the 3-month treatment, those who completed the treatment but without remission, and those who relapsed during follow-up.
Patients with moderate or severe depression could differ substantially in their health-related quality of life, so we also looked at health benefits in terms of quality-adjusted life-years (QALYs) gained. This generic outcome measure permits comparison of results between disease areas and also complies with current methodological guidance by NICE (Reference Richardson, Wonderling and SimonsRichardson et al, 2004). Quality-adjusted life-years were calculated using the combination of quality-of-life weights with the estimated length of time patients spend in the corresponding health states through the different clinical pathways in the decision model. The quality-of-life weights were obtained by a systematic review of the economic evidence of depression. Details of the economic literature review are reported elsewhere (National Collaborating Centre for Mental Health, 2005). Here we present only identified evidence relevant to the current study. Table 1 lists all the clinical effectiveness estimates and quality-of-life weights used in the decision analysis.
Costs and cost-effectiveness
Since no patient-level data were available to calculate costs in the model, deterministic costing of the different treatment strategies was carried out. Costs were identified from the perspective of the NHS and expressed in UK £ at 2002–3 prices. We calculated the cost of the initial treatment protocols (medication costs, staff costs, dispensing fees) using resource use estimates based on both the expert opinion of the Guideline Development Group and the literature. The cost of the likely subsequent healthcare resource use for the 15-month analysis period was based on estimates obtained from the systematic review of the economic evidence.
The antidepressant therapy protocol consisted of 3 months of daily 40 mg fluoxetine (British Medical Association & Royal Pharmaceutical Society of Great Britain, 2003), prescribed as part of standard outpatient care with an average of four specialist visits, assuming two visits by a consultant and two visits by a specialist registrar (expert opinion of the Guideline Development Group). Fluoxetine was advocated as the best antidepressant to represent routine pharmacotherapy for patients with moderate or severe depression because, as with other selective serotonin reuptake inhibitors, it has low toxicity in overdose compared with other classes of antidepressant (Reference Freemantle, House and SongFreemantle et al, 1994). It was also the most widely prescribed antidepressant in 2002 in England (Department of Health, 2003).
Combination therapy was defined as the combined use of the antidepressant treatment protocol (described above) and cognitive–behavioural therapy for a 3-month period. A full course of cognitive–behavioural therapy included 16 sessions, each session lasting for an average of 50 min (Reference McCulloughMcCullough, 1995). Based on the expert opinion of the Guideline Development Group, clinical psychologists were used as the most representative example of therapists providing this treatment in the UK. Such a treatment strategy was also in line with the treatment protocols of the reviewed clinical studies.
To calculate the cost of the initial treatment, we combined the estimated resource use with UK-specific unit costs obtained from the BNF (British Medical Association & Royal Pharmaceutical Society of Great Britain, 2003), the unit cost dictionary of the Personal Social Services Research Unit (Reference Netten and CurtisNetten & Curtis, 2002) and the Prescription Pricing Authority (http://www.ppa.gov.uk). Although occasionally missed treatment sessions meant that full costs were incurred, treatment costs were revised to account for patients who did not complete the treatment. For this, it was assumed that patients who discontinue initial therapy have on average 3 weeks of treatment (Reference Elkin, Shea and WatkinsElkin et al, 1989; expert opinion of the Guideline Development Group). Patients in remission who do not relapse during the 12-month follow-up have no subsequent treatment associated with depression. Patients with unsuccessful treatment outcomes, however, consume additional healthcare resources as part of their depression management over the analysis time horizon. Cost data for the subsequent depression treatment were taken from published research (Reference Borghi and GuestBorghi & Guest, 2000). These subsequent depression treatment costs included hospitalisation; emergency department, out-patient and general practitioner visits; community psychiatric nurse and community mental health team visits; and medication costs (Reference Borghi and GuestBorghi & Guest, 2000). The original estimate was adjusted to 2002–3 prices using the Hospital and Community Health Services inflation index (Reference Netten and CurtisNetten & Curtis, 2003) and projected for the periods during which unsuccessfully treated patients would consume subsequent healthcare resources estimated in the model. The average relapse time after the end of initial treatment was 14 weeks (Reference Simons, Murphy and LevineSimons et al, 1986). All unit cost parameters used in the evaluation are reported in Table 1. Total costs of the different clinical pathways were calculated by adding the initial treatment and subsequent healthcare resource use costs. Since the analysis time horizon was 15 months, discounting was not performed either for costs or for outcomes as it would not have had a significant effect on the results.
The overall clinical and cost outcomes were synthesised in the decision analysis. Incremental cost-effectiveness ratios in terms of ‘cost per additional successfully treated patient’ and ‘cost per QALY gained’ were estimated by dividing the difference in the expected total healthcare costs of the two strategies by the difference in their overall effects (Table 2).
Table 2 Expected costs, effects and cost-effectiveness results over 15 months
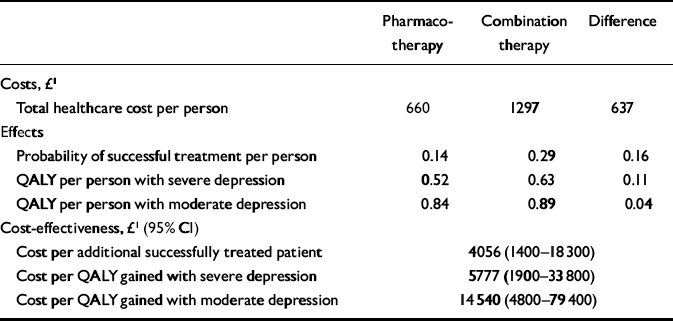
| Pharmacotherapy | Combination therapy | Difference | |
|---|---|---|---|
| Costs, £1 | |||
| Total healthcare cost per person | 660 | 1297 | 637 |
| Effects | |||
| Probability of successful treatment per person | 0.14 | 0.29 | 0.16 |
| QALY per person with severe depression | 0.52 | 0.63 | 0.11 |
| QALY per person with moderate depression | 0.84 | 0.89 | 0.04 |
| Cost-effectiveness, £1 (95% CI) | |||
| Cost per additional successfully treated patient | 4056 (1400-18 300) | ||
| Cost per QALY gained with severe depression | 5777 (1900-33 800) | ||
| Cost per QALY gained with moderate depression | 14 540 (4800-79 400) | ||
Sensitivity analyses
Policy implications of cost-effectiveness point estimates are ambiguous. We treated the national unit cost data and assumptions of the cognitive–behavioural therapy protocol as fixed parameters, but extensive sensitivity analysis was carried out to explore the uncertainty around the different input values and assumptions taken from the literature or based on the expert opinion of the Guideline Development Group. First, a univariate sensitivity analysis was carried out whereby single parameters were varied between their plausible minimum and maximum values while keeping all the other variables at their base case values (Reference Briggs and SculpherBriggs & Sculpher, 1995). We looked separately at the uncertainty around the various risk difference estimates, quality-of-life weights and the likely cost of subsequent depression treatment. We also explored the scenario of intensive clinical management of antidepressant therapy (Reference Fawcett, Epstein and FiesterFawcett et al, 1987)–additional care over and above what would usually be provided in routine care by the NHS–since most of the trials from which the clinical effectiveness estimates were derived administered trial medication using this protocol. Recent clinical recommendations also support a more intensive management of antidepressant therapy than current standard practice for patients with severe forms of depression (National Institute for Clinical Excellence, 2004). We also investigated the effect of adjusting for partial quality-of-life improvement in treatment-completer patients with no remission. Table 3 presents the ranges over which the different parameters were tested.
Table 3 Univariate sensitivity analysis
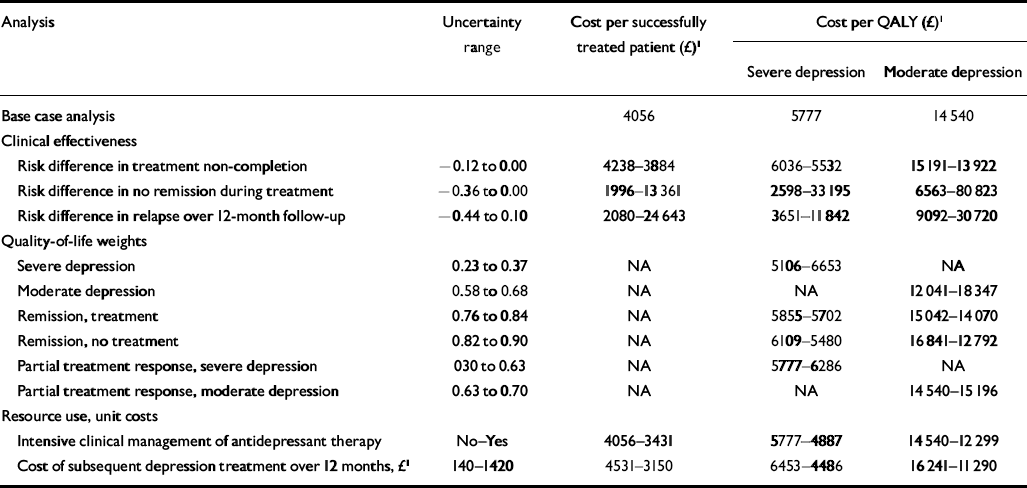
| Analysis | Uncertainty range | Cost per successfully treated patient (£)1 | Cost per QALY (£)1 | |
|---|---|---|---|---|
| Severe depression | Moderate depression | |||
| Base case analysis | 4056 | 5777 | 14 540 | |
| Clinical effectiveness | ||||
| Risk difference in treatment non-completion | -0.12 to 0.00 | 4238-3884 | 6036-5532 | 15 191-13 922 |
| Risk difference in no remission during treatment | -0.36 to 0.00 | 1996-13 361 | 2598-33 195 | 6563-80 823 |
| Risk difference in relapse over 12-month follow-up | -0.44 to 0.10 | 2080-24 643 | 3651-11 842 | 9092-30 720 |
| Quality-of-life weights | ||||
| Severe depression | 0.23 to 0.37 | NA | 5106-6653 | NA |
| Moderate depression | 0.58 to 0.68 | NA | NA | 12 041-18 347 |
| Remission, treatment | 0.76 to 0.84 | NA | 5855-5702 | 15 042-14 070 |
| Remission, no treatment | 0.82 to 0.90 | NA | 6109-5480 | 16 841-12 792 |
| Partial treatment response, severe depression | 030 to 0.63 | NA | 5777-6286 | NA |
| Partial treatment response, moderate depression | 0.63 to 0.70 | NA | NA | 14 540-15 196 |
| Resource use, unit costs | ||||
| Intensive clinical management of antidepressant therapy | No—Yes | 4056-3431 | 5777-4887 | 14 540-12 299 |
| Cost of subsequent depression treatment over 12 months, £1 | 140-1420 | 4531-3150 | 6453-4486 | 16 241-11 290 |
To demonstrate the joint uncertainty between the different parameters and to estimate the 95% uncertainty interval around the cost-effectiveness ratios, a probabilistic analysis was conducted. Using the mean parameter values and their 95% confidence intervals (see Table 1), appropriate distributions were assigned for each parameter estimate. For example, beta distributions were used for probabilities and lognormal distributions for costs. The outcome and cost estimates were then recalculated 10 000 times using Monte Carlo simulation (Reference Briggs, Goeree and BlackhouseBriggs et al, 2002). Whether an intervention is cost-effective or not depends on how decision-makers value the additional health gain achieved by the treatment. The probability that combination therapy is cost-effective compared with antidepressant therapy alone as a function of decision-makers’ maximum willingness to pay for an additional successfully treated patient or QALY was illustrated by acceptability curves (Reference Briggs and GrayBriggs & Gray, 1999).
RESULTS
Outcomes
Twenty-nine studies were included in the original clinical systematic review of cognitive–behavioural therapy (National Collaborating Centre for Mental Health, 2005). Out of these, seven trials compared individual cognitive–behavioural therapy plus antidepressants with antidepressant therapy alone in 831 patients with moderate or severe depression and provided data on numbers not completing treatment: Blackburn et al (Reference Blackburn, Bishop and Glen1981), Murphy et al (Reference Murphy, Simons and Wetzel1984), Miller et al (Reference Miller, Norman and Keinter1989), Hautzinger et al (Reference Hautzinger, de Jong-Meyer and Treiber1996), Scott et al (Reference Scott, Tacchi and Jones1997), Keller et al (Reference Keller, McCullough and Klein2000) and Thompson et al (2001). Four studies provided data on remission for 646 patients with moderate or severe depression at the end of treatment: Murphy et al (Reference Murphy, Simons and Wetzel1984), Elkin et al (Reference Elkin, Shea and Watkins1989), Miller et al (Reference Miller, Norman and Keinter1989) and Keller et al (Reference Keller, McCullough and Klein2000). Subsequent reports of two trials provided data on outcomes during 12-month follow-up: Blackburn et al (Reference Blackburn, Euson and Bishop1986) and Simons et al (Reference Simons, Murphy and Levine1986); the latter reference is a follow-up report of the trial by Murphy et al (Reference Murphy, Simons and Wetzel1984).
The probability of not completing 3-month treatment was lower (risk difference (RD)= –0.06, 95% CI –0.12 to 0.00) and the probability of not achieving remission during treatment was significantly reduced (RD= –0.18, 95% CI –0.36 to 0.00) with combination therapy compared with antidepressant medication alone (Figs 2 and 3). The follow-up studies suggested that there is also lower risk of relapse with combination therapy (RD= –0.17, 95% CI –0.44 to 0.1) over a 12-month follow-up (Fig. 4). The decision analysis showed an overall 0.16 increase per patient in the probability of successful treatment at the end of the 15-month analysis time horizon with combination therapy (see Table 2).
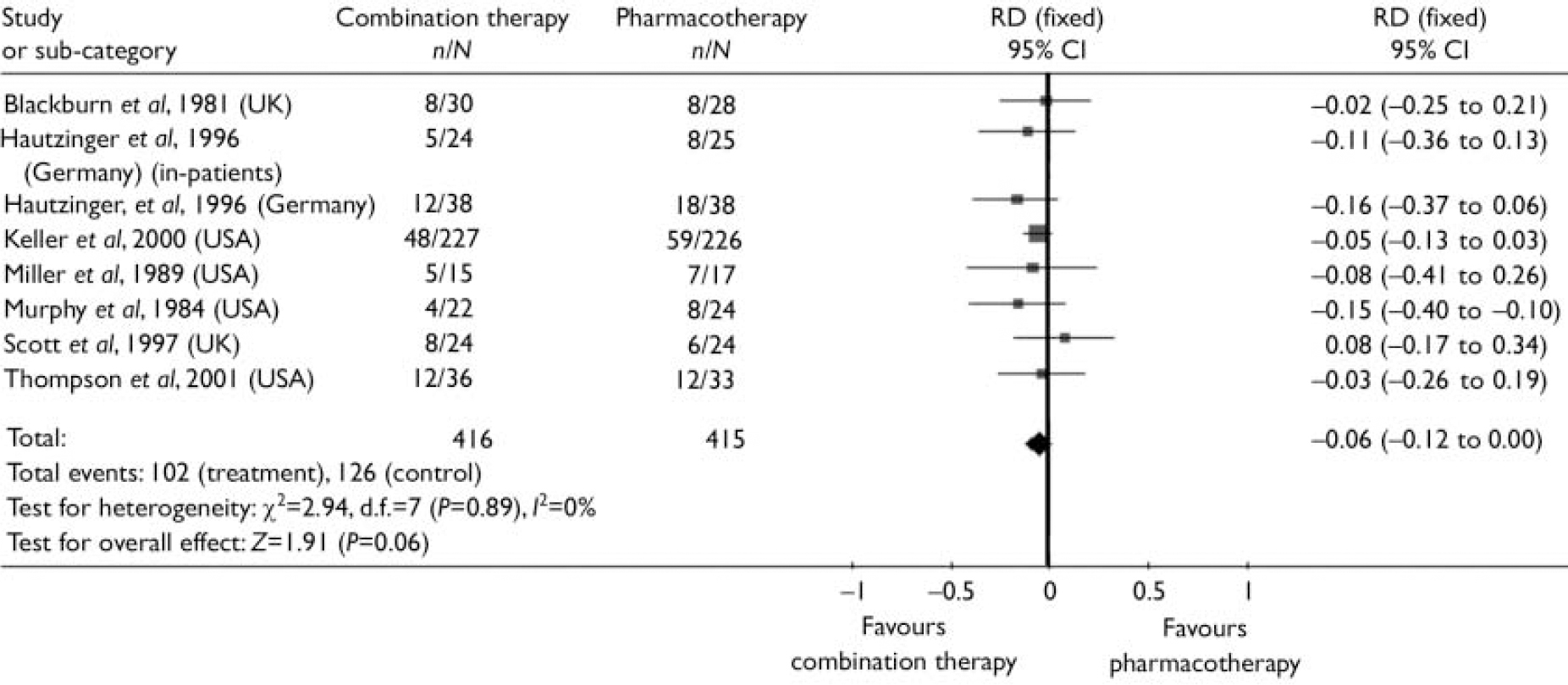
Fig. 2 Risk difference (RD) between combination therapy and pharmacotherapy for moderate and severe depression: patients not completing 3-month treatment.
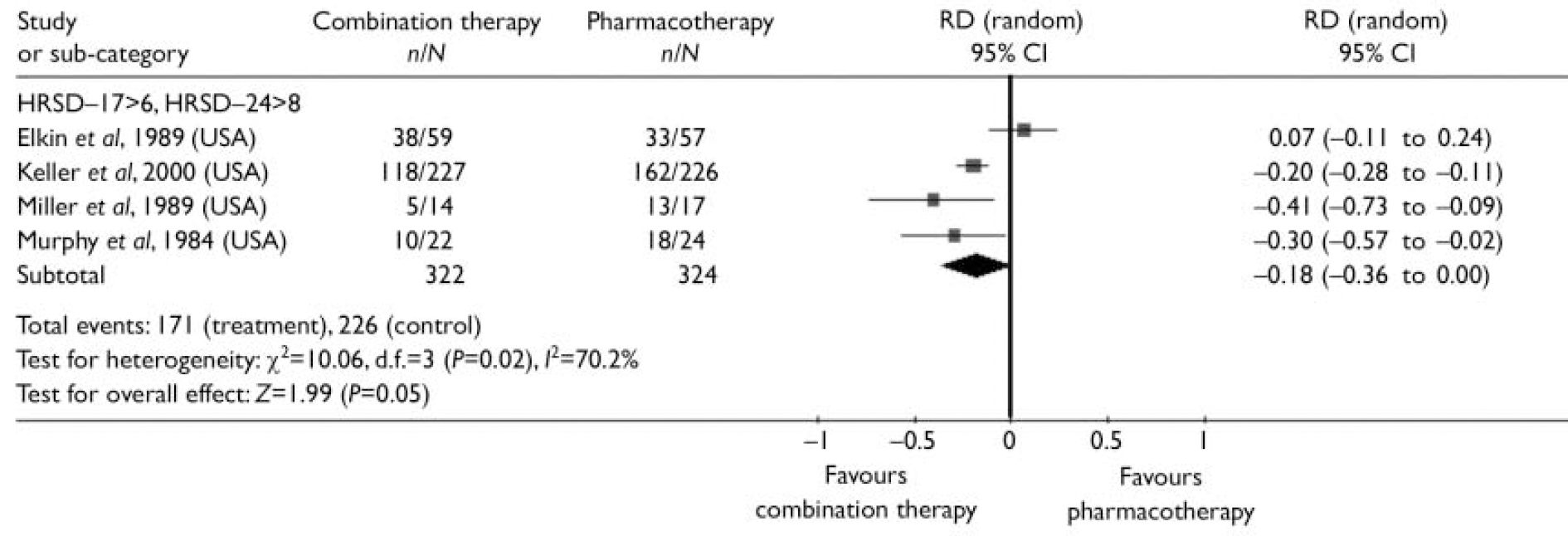
Fig. 3 Risk difference (RD) between combination therapy and pharmacotherapy for moderate and severe depression: patients not achieving remission during treatment.
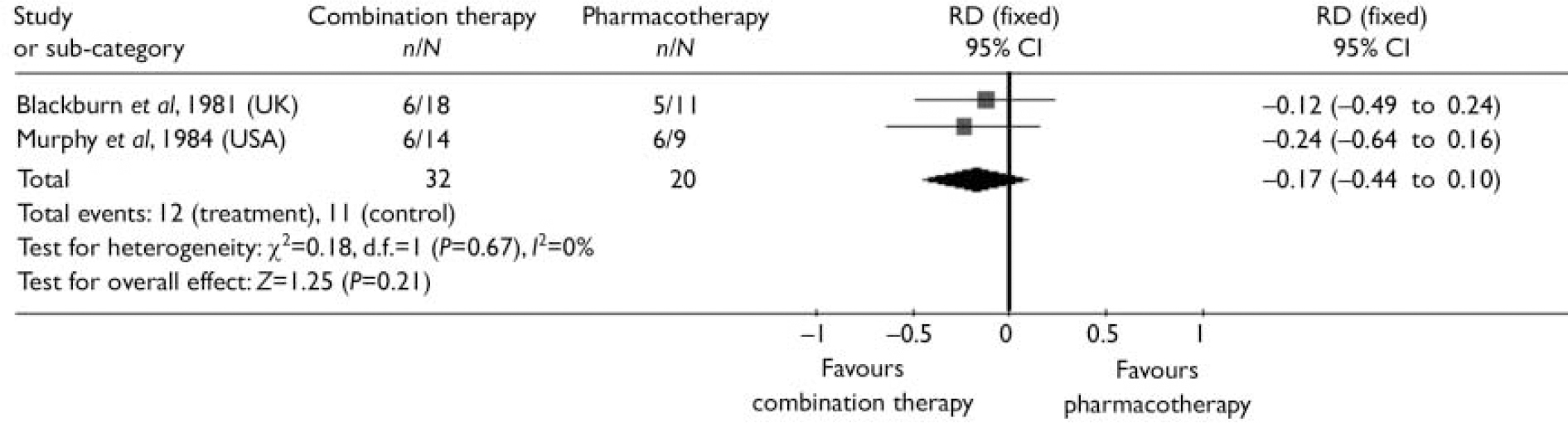
Fig. 4 Risk difference (RD) between combination therapy and pharmacotherapy for moderate and severe depression: relapse over 12-month follow-up.
The economic literature review identified only one study with data on quality-of-life weights appropriate for this decision analysis. Revicki & Wood (Reference Revicki and Wood1998) elicited patient-assigned health state utilities by standard gamble technique for a variety of depression severity and different treatment statuses with fluoxetine. Using these estimates, the average gain in QALYs was calculated at 0.11 per patient with severe depression and 0.04 per patient with moderate depression over the 15-month analysis period (see Table 2).
Costs and cost-effectiveness
The full cost of a 3-month course of anti-depressant therapy with standard care was estimated as £162 at 2002–3 prices. The expected subsequent healthcare cost of managing someone with moderate or severe depression who did not respond to initial treatment was £580 for the analysis period. The expected cost of relapse within the analysis time horizon was £417. The initial treatment cost of combination therapy was £1029 including the cost of a full course of cognitive–behavioural therapy estimated at £867. Taking into consideration likely savings in subsequent healthcare use by combination therapy, it is estimated that overall combination therapy would cost an extra £637 per patient when used for the routine treatment of moderate or severe depression in specialist care during the analysis period. The cost-effectiveness of combination therapy was calculated at £4056 per additional successfully treated patient, resulting in a cost of £5777 per QALY gained for severe depression and £14 540 per QALY gained for moderate depression (see Table 2).
Sensitivity analyses
Table 3 reports the results of the univariate sensitivity analysis. This indicates that the results are robust to the investigated input parameters and assumptions. The cost-effectiveness estimates are most sensitive to the uncertainty around the difference in the risk of no remission at the end of treatment between the two treatment strategies. Other factors have a much lesser role in the variation of the results.
The probabilistic analysis showed that the 95% confidence interval around the cost per additional successfully treated patient is between £1400 and £18 300. When taking patients’ quality of life into consideration, there is 97% probability that combination therapy is more cost-effective than antidepressant therapy alone for severe depression (95% CI £1900–33 800 per QALY) and 88% probability for moderate depression (95% CI £4800–79 400 per QALY) at a recently quoted £30 000 as decision-makers’ maximum willingness to pay per QALY in the UK (Reference Richardson, Wonderling and SimonsRichardson et al, 2004). In contrast to severe depression, however, the probability of cost-effectiveness for moderate depression is greatly affected by the maximum willingness to pay value. At values lower than £30 000 per QALY, the uncertainty around the cost-effectiveness of combination therapy greatly increases for patients with moderate depression (Fig. 5).
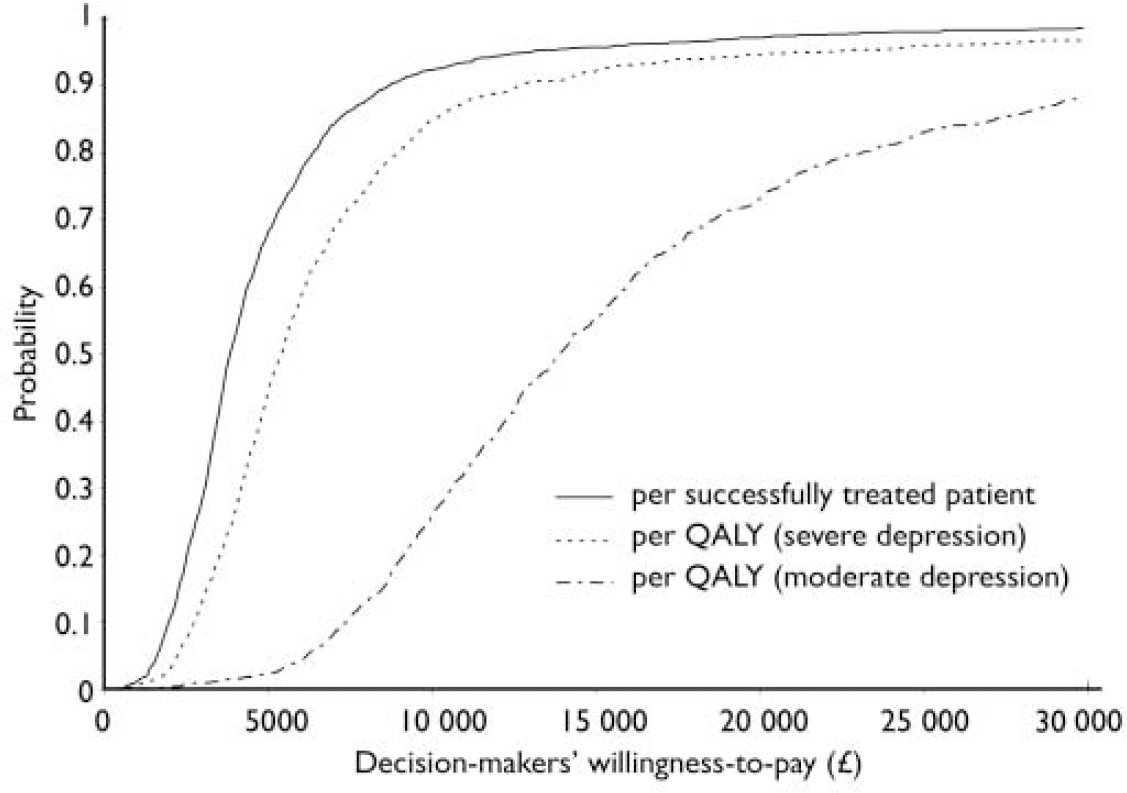
Fig. 5 Cost-effectiveness acceptability curves: probability that combination therapy is cost-effective as a function of decision-makers’ willingness to pay per additional unit of health gain. QALY, quality-adjusted life-year.
DISCUSSION
Assessment of the number of successfully treated patients reveals an additional benefit of cognitive–behavioural therapy in combination with antidepressant therapy over pharmacotherapy alone which is similar for both moderate and severe depression. However, the decision analysis predicts that when patients’ quality of life is also included, there are greater gains for patients with severe depression compared with moderate depression. The cost results suggest that although the initial treatment cost of combination therapy is substantially higher, these costs are in part offset by savings accruing from lower treatment costs in the subsequent year. Overall, the evaluation shows that combination therapy is likely to be a cost-effective first-line secondary care treatment for severe depression. It is, however, much more uncertain from the currently available evidence whether its first-line secondary care use is cost-effective for moderate depression. Following from these results, it is also likely that targeting combination therapy on severe forms of depression could be a more efficient way of using limited resources.
Generalisability of the results
Although the cost-effectiveness estimates were based on costs from the UK, the effectiveness data were synthesised from a range of different countries and healthcare systems. In the case of high-income countries, the relative costs and benefits of the different interventions (in particular the psychological treatment) are expected to be similar. Therefore the findings of this study are likely to be generalisable across high-income countries. However, different countries have different thresholds for society's maximum willingness to pay per additional health benefit, which may affect the decision to provide combination therapy as first-line treatment for either moderate or severe depression.
Limitations of the analysis
In our study clinical effectiveness estimates were based on efficacy data obtained from randomised controlled trials, and so it is possible that the probability of successful outcome is overestimated for both treatment options provided within standard NHS care. It is anticipated, however, that this does not influence the relative effectiveness of the compared interventions significantly. Since a number of the included studies were conducted during the 1980s, issues may also arise regarding current applicability of the synthesised clinical evidence. The results of more recent studies (for example, Reference Keller, McCullough and KleinKeller et al, 2000) suggest that this is not the case.
As with previous studies, we defined successful treatment as achieving remission at the end of 3-month treatment and no relapse during 12-month follow-up, but in the sensitivity analysis we also investigated the effect of partial response to treatment when quality of life was included. Current evidence on the health-related quality of life of people with depression, however, is very scarce. Therefore, revision of all the QALY estimates will be necessary when more utility information becomes available. Furthermore, some clinical evidence also indicates lower relapse rates with combination therapy for up to 6 years after treatment (Reference Scott, Palmer and PaykelScott et al, 2003). It would have been desirable to evaluate the different strategies over a longer period but we were unable to extend our evaluation this far because of lack of direct clinical evidence beyond 15 months.
Regarding the limitations of the cost difference estimates, individual cognitive–behavioural therapy is a relatively resource-intensive psychological treatment. In future, clinical research may define and develop less resource-intensive but similarly effective forms of psychological therapy to be used in combination with anti-depressants, thereby improving cost-effectiveness. It is unlikely that the choice of antidepressant would have influenced the cost difference between the combination and the pharmacotherapy strategies substantially, since the assumed medication protocol is identical in the two strategies. Similar reasoning is likely to be valid for the cost consequences of maintenance anti-depressant therapy over and above the initial 3-month treatment. This option was not tested because of the lack of appropriate comparative clinical evidence.
Depression incurs significant non-healthcare costs such as social service costs, direct costs to patients and their families and lost productivity costs due to morbidity or premature mortality (Reference Knapp and IlsonKnapp & Ilson, 2002). As the present analysis was carried out from the perspective of the healthcare sector, these non-healthcare costs were not considered. Had we included them, it is likely that the cost-effectiveness of combination therapy would have improved. All the described limitations of the clinical and cost difference evidence suggest that current results of the decision model should be rather treated as conservative.
Guideline recommendations
Based on the clinical and cost-effectiveness evidence and the availability and likely affordability of the different treatment options within the NHS, the NICE guideline (National Institute for Clinical Excellence, 2004) recommends combination therapy when patients present initially with severe depression. Antidepressants are the recommended first-line treatment for those with moderate depression, with combination therapy as second-line intervention. Considering the importance of patient preference, recommendations are also made about the use of psychological interventions, including combination treatment, where patients declined the offer of anti-depressant medication alone or had previously not responded to antidepressant medication. The consequent increase in the uptake of psychological treatments for people with severe depression and moderate depression that has not responded to antidepressants is likely to require additional resources in the NHS, mostly the cost and time implications of extra staff training and employment. These are currently under consideration by NICE and the NHS in England and Wales.
Acknowledgements
We thank members of the Depression Guideline Development Group (D. Goldberg, S. Pilling, T. Kendall, N. Ferrier, T. Foster, J. Gates, P. Gilbert, P. Harvey, I. Hughes, C. Paton, S. Rippon, K. Sheldon, D. Turkington, A. Tylee) for their expert advice on the decision analysis undertaken to support the guideline, especially Paul Gilbert, University of Derby, and Ian Hughes, Cardiff and Vale NHS Trust, for acting as clinical reviewers. We also thank Preethi Premkumar and Lisa Underwood, National Collaborating Centre for Mental Health, for assisting with the data extraction for the clinical effectiveness review. We are grateful to the referees for their useful comments on the manuscript.
The guideline development process was commissioned and funded by the UK National Institute for Health and Clinical Excellence. Judit Simon is funded by a Research Scientist in Evidence Synthesis award from the Department of Health and NHS Research and Development, UK. The authors are grateful for all their support.













eLetters
No eLetters have been published for this article.