Cognitive–behavioural therapy (CBT) for chronic fatigue syndrome is an effective Reference Chambers, Bagnall, Hempel and Forbes1 but intensive treatment. It requires 13–16 sessions, depending on the protocol used. Reference Deale, Chalder, Marks and Wessely2–Reference Prins, Bleijenberg, Bazelmans, Elving, de Boo, Severens, van der Wilt, Spinhoven and van der Meer4 We assumed that for a subgroup of patients with chronic fatigue syndrome a less intensive treatment sufficed and developed a minimal intervention based on CBT that consisted of self-instructions and email contact. The effects of self-instructions on fatigue and disabilities were compared with a waiting-list condition in a randomised controlled trial. The predictive value of the level of fatigue and disabilities at baseline for treatment outcome was determined.
Method
Patients referred for CBT to our tertiary care facility were eligible to enter the study if they were ⩾18 years old; spoke and read Dutch; met the 1994 US Center for Disease Control and Prevention criteria for chronic fatigue syndrome; Reference Fukuda, Straus, Hickie, Sharpe, Dobbins and Komaroff5 were not engaged in a legal procedure concerning disability-related financial benefits; Reference Prins, Bazelmans, van der Werf, van der Meer, Bleijenberg, Sivik, Byrne, Lipsitt, Cristodoulou and Dienstfrey6 scored ⩾35 on the Checklist Individual Strength (CIS), Reference Vercoulen, Swanink, Fennis, Galama, van der Meer and Bleijenberg7 fatigue severity sub-scale; had a total score of >700 on the Sickness Impact Profile–8 (SIP8); Reference Knoop, Bleijenberg, Gielissen, van der Meer and White8 and had given written informed consent. The local ethics committee approved the study.
After baseline assessment performed by research assistants, patients were offered CBT. If they agreed, they were placed on a waiting list for a period of 6–12 months depending on available treatment capacity. Patients were informed about the study and if they gave informed consent, were randomly assigned to either the guided self-instructions or the waiting-list condition. Allocation to group was carried out by a therapist using cards in consecutively numbered opaque and sealed envelopes that were opened in the presence of the patient. A researcher checked every week whether the sequence was subverted by matching the date the patient was included with the sequence of the numbers and date of the session. No irregularities were found. A statistical advisor prepared the envelopes by coding them according to a computer-generated list of random numbers in blocks of eight. Patients were assessed at baseline and directly following the waiting period or intervention. This could vary between 6 and 12 months depending on the available treatment capacity.
The intervention consisted of a self-instruction booklet containing information about chronic fatigue syndrome and weekly assignments. The programme took at least 16 weeks, but often more if patients formulated long-term goals such as returning to work. Patients were asked to email (or telephone if they did not have email) at least once every 2 weeks to report their progress. A cognitive–behavioural therapist, trained in regular CBT for chronic fatigue syndrome, responded to this email or call. If patients did not respond every 2 weeks, a reminder was sent by email or patients were telephoned.
The CIS sub-scale ‘fatigue severity’ was used to measure the level of fatigue over the past 2 weeks. Scores ranged from 8 (no fatigue) to 56 (severely fatigued). Reference Vercoulen, Swanink, Fennis, Galama, van der Meer and Bleijenberg7 The weighted total score on eight sub-scales of the SIP8 (SIP8 total score) was used to assess functional disability in all domains of functioning. Reference Knoop, Bleijenberg, Gielissen, van der Meer and White8 Physical disabilities were measured with the physical functioning sub-scale of the 36-item Short Form Health Survey (SF–36). Reference Stewart, Hays and Ware9 Scores ranged from 0 (maximum physical limitations) to 100 (ability to do vigorous activity).
Clinically significant improvement Reference Jacobsen and Truax10 was defined as a reliable change index >1.96 and a score of <35 on the CIS sub-scale fatigue severity at second assessment. This score is within two standard deviations of the mean for healthy adults. Reference Knoop, Bleijenberg, Gielissen, van der Meer and White8 A score of <35 would reflect a significant reduction of fatigue. We assumed that in the waiting-list condition 10% of the patients would have a fatigue score of <35. Power calculation showed that 98 patients in each condition were needed to detect a difference of 15% in the proportion of patients with a fatigue score within normal limits, assuming a significance of 5%, power of 90% and a drop-out rate of 20%.
Data analyses were performed using SPSS (version 14) for Windows. Significance was assumed at P<0.05. To test whether there was a difference between the two conditions on outcome measures, ANCOVA Reference Van Breukelen11 was used with the score on the second assessment as the dependent variable, baseline score as the covariate and condition as the fixed factor. To test whether the proportion of patients with a clinically significant improvement differed between conditions, logistic regression with condition and baseline CIS fatigue as predictors was used. To test whether the treatment effect was moderated by patient characteristics, ANCOVA for CIS fatigue on the second assessment was repeated with condition (standardised; Z-score with mean=0, s.d.=1), baseline CIS fatigue (standardised), baseline SIP8 total score, condition×baseline CIS score, and condition×baseline SIP8 score as predictors. All comparisons were performed on the basis of intention to treat. For missing data, the last observation was carried forward.
Results
During the study it became clear that the number of patients lost at the second assessment was lower than expected (about 5%). The power analysis was repeated but now using an estimated drop-out rate of 5%, indicating that a sample size of 85 in each condition sufficed. The inclusion of patients stopped when this sample size was reached (Fig.1).
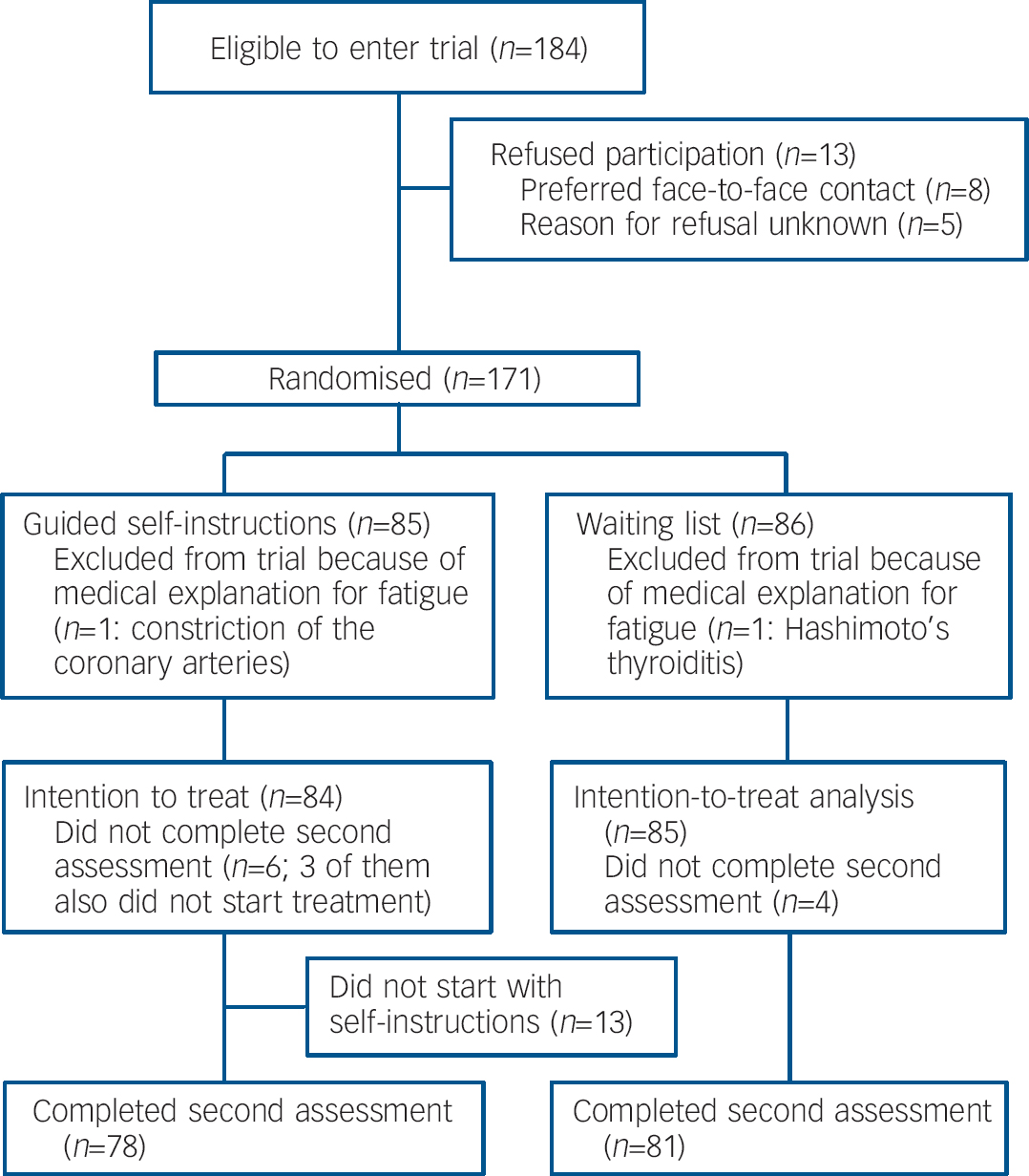
Fig. 1 Flow of participants through the study.
Of 84 patients in the self-instructions condition, 55 (66%) emailed, 5 (6%) exclusively used the telephone and 10 (12%) did both. Fourteen patients (16%) had no contact with a therapist: 1 patient completed the programme by herself, the remaining 13 did not start. There was no significant difference in mean time passed in months between the two assessments for the guided self-instruction (10.5 months, s.d.=4.0) and the waiting-list condition (9.7 months, s.d.=3.6, t=1.34, d.f.=157, P=0.182). Patients from the intervention condition were significantly less fatigued (intervention mean=38.9 v. waiting-list mean=46.4), reported fewer disabilities (mean=1079 v. mean=1319), scored significantly higher on the SF–36 physical functioning sub-scale (mean=65.9 v. mean=60.2) and more often showed a clinically significant improvement in fatigue (27% v. 7%) at second assessment (online Tables DS1–3).
Of the interaction terms, only the condition×SIP8 total score interaction effect was significant (B=3.533, t=2.250, P=0.026), indicating that the treatment effect is more than halved for patients with an SIP8 score of 1 standard deviation above the mean.
Discussion
It was already known that individual CBT is an effective treatment for chronic fatigue syndrome. Reference Chambers, Bagnall, Hempel and Forbes1 Our study showed that a less intensive intervention based on the same principles also leads to a decrease of fatigue and disabilities in patients with chronic fatigue syndrome. For the subgroup of patients that showed a clinically significant improvement in fatigue, the minimal intervention sufficed. Self-instructions could form the first step in a model of stepped care for chronic fatigue syndrome. The results also suggest that more severely disabled patients benefit less from the self-instructions and could perhaps be referred for face-to-face CBT instead.
As we did not use a control condition we cannot be sure that the specific elements in the minimal intervention condition were responsible for the reduction of fatigue and disabilities. However, two randomised controlled trials that compared the effect of CBT for chronic fatigue syndrome with a placebo or non-specific condition both showed CBT to have a superior effect. Reference Deale, Chalder, Marks and Wessely2,Reference Prins, Bleijenberg, Bazelmans, Elving, de Boo, Severens, van der Wilt, Spinhoven and van der Meer4 Furthermore, Cho et al Reference Cho, Hotopf and Wessely12 showed that the placebo response of patients with chronic fatigue syndrome to psychological interventions is lower than in other medical conditions.
Acknowledgements
Trial register number: ISRCTN27293439. Ethical approval was obtained from the human ethics committee of the Radboud University Nijmegen Medical Centre. We thank the patients who participated in the study and Thea Berends, Annemarie Gerritsen, Gerrie van de Heijdt, Henriette Vermeer and Hein Voskamp for treating the patients in the guided self-instruction condition, Carel Kruip, Tiny Fasotti and Lianne Vermeer for their assistance in data collection, and Rogier Donders for statistical advice.






eLetters
No eLetters have been published for this article.