Early accurate diagnosis of dementia with Lewy bodies, especially its differentiation from Alzheimer's disease, is important for optimum management, including provision of appropriate information to patients and carers, initiation of effective treatments and avoidance of potentially life-threatening antipsychotic drugs. Reference McKeith, Dickson, Lowe, Emre, O'Brien and Feldman1 Consensus clinical diagnostic criteria have high specificity for diagnosis of probable dementia with Lewy bodies but poor sensitivity; Reference Litvan, Bhatia, Burn, Goetz, Lang and McKeith2 data for possible dementia with Lewy bodies are not available. Ancillary investigations, such as neuroimaging, may be important in enhancing accuracy of diagnosis. Nigrostriatal degeneration, accompanied by profound reductions in the striatal dopamine transporter is found in dementia with Lewy bodies. Reference Piggott, Marshall, Thomas, Lloyd, Court and Jaros3 It is possible to assess the dopamine transporter in vivo with specific ligands, such as [123I]-2β-carbomethoxy-3β-(4-iodophenyl)-N-(3-fluoropropyl)nortropane (123I-FP-CIT). Reference Booij, Habraken, Bergmans, Tissingh, Winogrodzka and Wolters4 A phase III multicentre study investigating 123I-FP-CIT SPECT (single photon emission computed tomography) in dementia with Lewy bodies reported a sensitivity of 77.7% for detecting probable dementia with Lewy bodies and a specificity of 90.4% for excluding non-Lewy body dementia (primarily Alzheimer's disease). Reference McKeith, O'Brien, Walker, Tatsch, Booij and Darcourt5 123I-FP-CIT SPECT has been incorporated into the revised diagnostic criteria Reference McKeith, Dickson, Lowe, Emre, O'Brien and Feldman1 and is recommended by the National Institute for Health and Clinical Excellence (NICE) for use to assist with the diagnosis of dementia with Lewy bodies (www.nice.org.uk).
Studies showing high diagnostic accuracy for 123I-FP-CIT SPECT Reference McKeith, O'Brien, Walker, Tatsch, Booij and Darcourt5–Reference Walker, Costa, Walker, Shaw, Gacinovic and Stevens7 have compared imaging against clinical diagnosis of probable dementia with Lewy bodies. Arguably, imaging investigations have greater potential to help clinicians where there is less diagnostic certainty – that is, in individuals with possible (one core feature – fluctuation, recurrent visual hallucinations or spontaneous parkinsonism) as opposed to probable (two or more core features) dementia with Lewy bodies. Studies in such groups require subsequent verification of diagnosis, either through autopsy or by further follow-up when disease progression clarifies the diagnosis.
We undertook a 12-month follow-up study of people with Lewy body or non-Lewy body dementia who had taken part in a previous study of 123I-FP-CIT SPECT in dementia, focusing on the outcome of the individuals with possible dementia with Lewy bodies who were not part of the primary outcome in the previous report. Reference McKeith, O'Brien, Walker, Tatsch, Booij and Darcourt5 Our aim was to assess the ability of 123I-FP-CIT SPECT to differentiate between people with Lewy body or non-Lewy body dementia with an initial uncertain clinical diagnosis of possible dementia with Lewy bodies.
Method
Participants
We undertook a 12-month follow-up assessment of participants previously enrolled to the McKeith et al phase III multicentre study. Reference McKeith, O'Brien, Walker, Tatsch, Booij and Darcourt5 For full inclusion criteria, please see that paper. Briefly, participants were aged 55–90 years, met the criteria for dementia detailed in DSM–IV and fulfilled at least one of the following: consensus criteria Reference O'Brien, Colloby, Fenwick, Williams, Firbank and Burn6 or NINCDS–ADRDA (National Institute of Neurological and Communicative Diseases and Stroke, and the Alzheimer's Disease and Related Disorders Association) criteria for probable or possible Alzheimer's disease, Reference McKhann, Drachman, Folstein, Katzman, Price and Stadlan8 or NINDS–AIREN (National Institute of Neurological Disorders and Stroke, and Association Internationale pour la Recherche et l'Enseignement en Neurosciences) criteria for probable or possible vascular dementia. Reference Roman, Tatemichi, Erkinjuntti, Cummings, Masdeu and Garcia9 A Mini-Mental State Examination (MMSE) score of 10 or more was required to ensure individuals could complete assessments. Reference Folstein, Folstein and McHugh10 Participants with dementia who developed parkinsonism more than 1 year before the onset of dementia were deemed to have Parkinson's disease with dementia and were not included. Reference McKeith, Galasko, Kosaka, Perry, Dickson and Hansen11 Those with structural imaging findings indicative of infarction in the region of the basal ganglia, including the internal capsule, were excluded. Use of medication known or suspected to interact with striatal binding of 123I-FP-CIT was not permitted.
The study was done in accordance with the current revision of the Declaration of Helsinki and the Good Clinical Practice: Consolidated Guideline approved by the International Conference on Harmonisation and applicable to national and local laws and regulations. At every participating site, the study protocol and all amendments were approved by an institutional review board or independent ethics committee. All participants and caregivers gave written informed consent.
Following inclusion in the initial study, we invited participants for re-assessment at 12 months.
Procedures
Clinical diagnosis at baseline, as previously reported, was established by an independent consensus panel of three specialist clinicians, who were provided with a participant profile compiled from quality-assured clinical data from the on-site investigators' case record forms and copies of on-site original source data. The same panel reconvened to consider the baseline and the 12-month follow-up data to arrive at a second and final consensus diagnosis.
The following were undertaken at baseline and follow-up: MMSE, Unified Parkinson's Disease Rating Scale (UPDRS) III (motor section), Reference Fahn12 modified Hoehn and Yahr staging, Reference Hoehn and Yahr13 Clinical Assessment of Fluctuation (CAF) scale, Reference Walker, Ballard, Ayre, Wesnes, Cummings and McKeith14 the Cambridge Cognitive Examination–revised version (CAMCOG–R), Reference Huppert, Brayne, Gill, Paykel and Beardsall15 Neuropsychiatric Inventory with caregiver input (NPI–D), Reference Cummings, Mega, Gray, Rosenberg-Thompson, Carusi and Gornbein16 Visual Object and Space Perception (VOSP) battery Reference Warrington and James17 and Clinical Dementia Rating (CDR). Reference Hughes, Berg, Danziger, Coben and Martin18 The Cornell Scale for Depression in Dementia Reference Alexopoulos, Abrams, Young and Shamoian19 and the investigator's estimation of the person's IQ level were completed at baseline, but not at follow-up. Results of magnetic resonance imaging and computed tomography scans and the on-site investigators' clinical diagnosis before imaging were also available. The consensus panel did not at any stage have access to 123I-FP-CIT SPECT findings and was unaware of the participants' identities, the names of the centre and the investigators. Before any participants were diagnosed, the consensus panel was asked to diagnose ten patients (separate to the study) for whom autopsy diagnosis was independently available. There was 100% concordance between the diagnoses made by the panel and at autopsy. Reference McKeith, O'Brien, Walker, Tatsch, Booij and Darcourt5 Individual panel members reviewed each study case, including the baseline consensus panel diagnosis and all subsequent information, before meeting to agree a final clinical diagnosis of probable dementia with Lewy bodies, possible dementia with Lewy bodies or non-Lewy body dementia. Participants in the non-Lewy body dementia category were further allocated to probable or possible Alzheimer's disease, probable or possible vascular dementia or other.
Within a few weeks of the baseline clinical diagnosis, SPECT images were acquired on a two- or three-headed camera 3–6 h after a single intravenous injection of 111-185 MBq of I-FP-CIT Reference Booij, Hemelaar, Speelman, de Bruin, Janssen and van Royen20 (the radiotracer was supplied by GE Healthcare). See McKeith et al for details. Reference McKeith, O'Brien, Walker, Tatsch, Booij and Darcourt5 Participants underwent standard thyroid blocking. Single photon emission computed tomography imaging was not repeated at follow-up. As previously described, three nuclear medicine physicians assessed scans, masked to diagnosis, using a 4-point scale (0, normal uptake; 1, unilateral putamen loss; 2, bilateral putamen loss; 3, virtually absent uptake); Reference McKeith, O'Brien, Walker, Tatsch, Booij and Darcourt5 we used only the dichotomous division of normal (0) v. abnormal (1–3) images for analysis.
Statistical analysis
We analysed data with SAS software (version 9.1) for Windows for binomially distributed data, using χ2 tests to assess differences among the diagnostic groups (probable dementia with Lewy bodies, possible dementia with Lewy bodies, non-Lewy body dementia) with respect to participants' characteristics. We used an analysis of variance (ANOVA) for normally distributed data; if normality could not be established, we used the non-parametric Kruskal–Wallis test.
Our aim was to compare the results of the visual assessment (normal or abnormal scan) in individuals with probable Lewy body or non-Lewy body dementia as determined by the final (12-month follow-up) consensus panel diagnosis. We were particularly interested in individuals reported by McKeith et al Reference McKeith, O'Brien, Walker, Tatsch, Booij and Darcourt5 to have possible dementia with Lewy bodies, and the ability of 123I-FP-CIT imaging at baseline to predict the final consensus panel diagnosis 12 months later.
Results
Figure 1 shows the participant profile. Of 325 participants invited for follow-up at 12 months, information was obtained for 264 (81%). Completer status varied slightly between diagnostic groups, with 129 of 147 (88%) in the non-Lewy body dementia group returning for follow-up compared with 72 of 94 (77%) in the probable dementia with Lewy bodies group and 46 of 57 (81%) in the possible dementia with Lewy bodies group. Of 28 (61%) participants with no initial diagnosis, 17 were followed-up. Age and gender did not differ between completers and non-completers. Of 264 participants who completed the 12-month follow-up assessment, 29 were excluded from the analysis (Fig. 1).
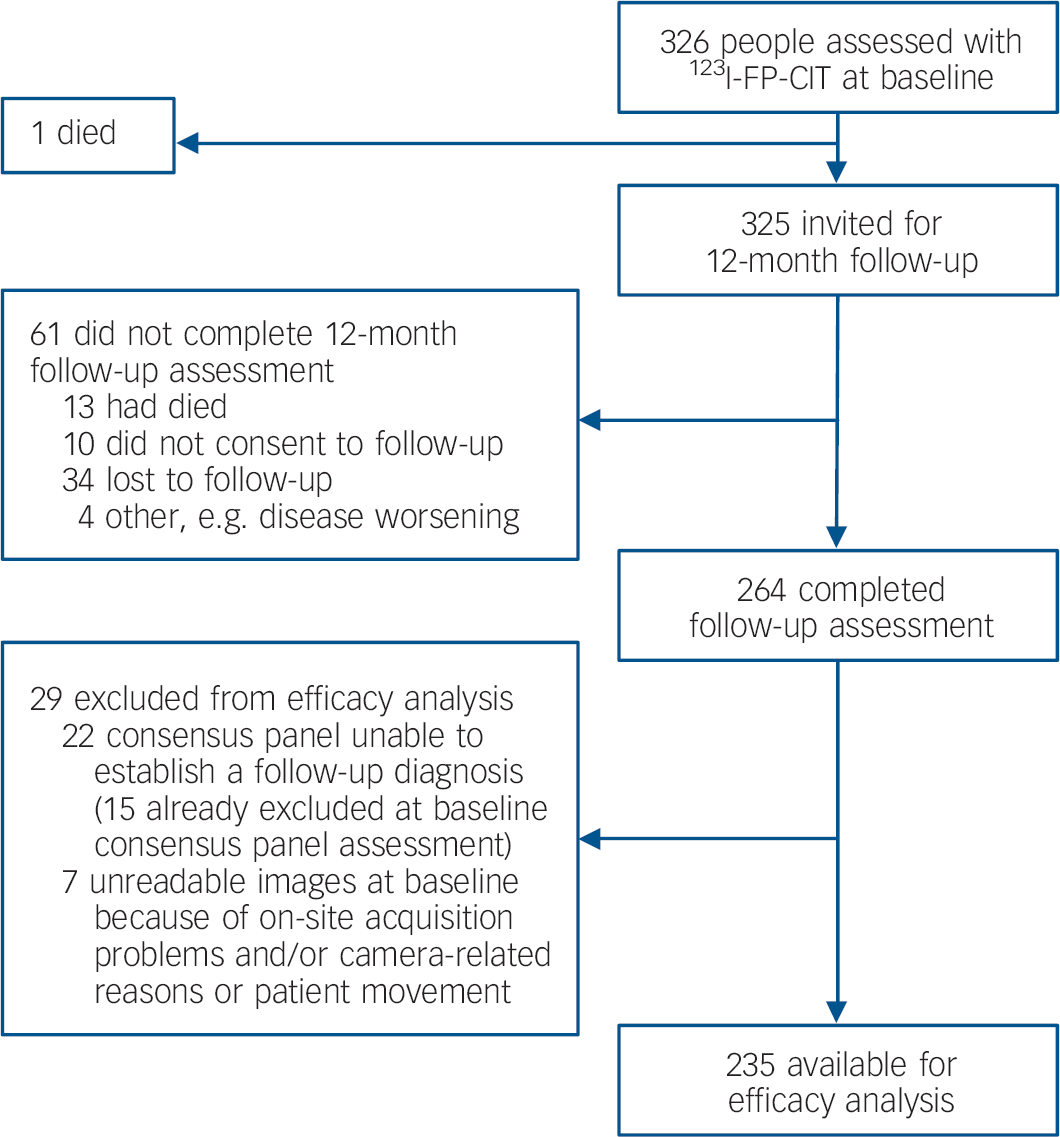
Fig. 1 Trial profile.
Follow-up diagnoses for the 235 participants available for the efficacy analysis were probable dementia with Lewy bodies (n=86), possible dementia with Lewy bodies (n=25) and non-Lewy body dementia (n=124; of which 84 were diagnosed as probable Alzheimer's disease, 29 possible Alzheimer's disease, 1 probable vascular dementia, 9 possible vascular dementia and 1 dementia post-surgery).
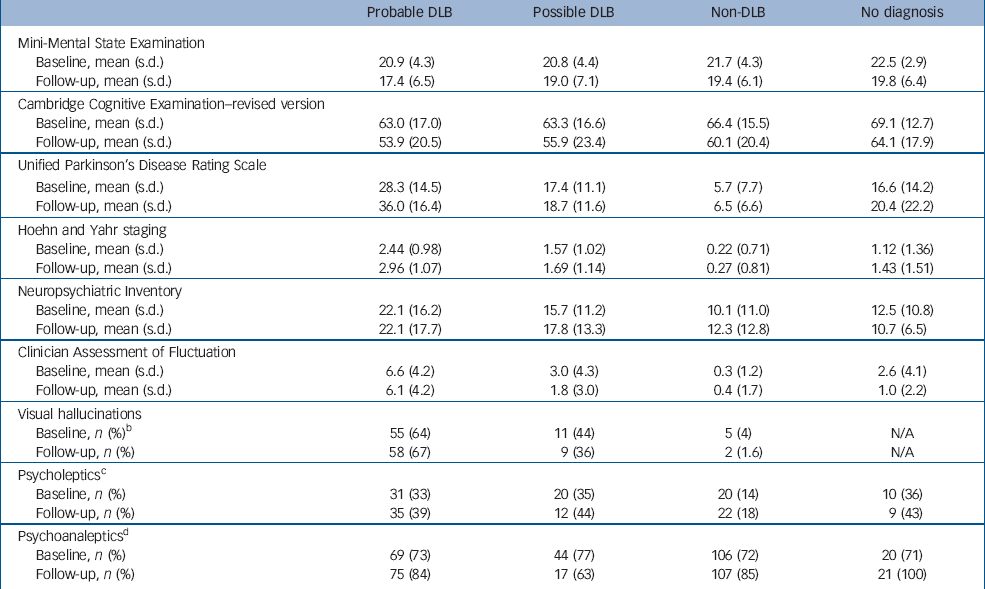
Diagnostic groups at follow-up were similar in terms of mean age (probable dementia with Lewy bodies, 73.7 years; possible dementia with Lewy bodies, 76.0; non-Lewy body dementia, 74.2; no diagnosis, 69.8) with, as expected, more men in the dementia with Lewy bodies groups (percentage of men with probable dementia with Lewy bodies, 62%; possible dementia with Lewy bodies, 67%; non-Lewy body dementia, 50%; no diagnosis, 64%). Table 1 shows the clinical characteristics of the groups at baseline and follow-up. Groups were well matched for baseline MMSE and CAMCOG; participants in all three dementia groups showed evidence of cognitive decline over 12 months. Patients with dementia with Lewy bodies also showed evidence of worsening motor symptoms over the follow-up period. Behavioural disturbance – as judged by the neuropsychiatric inventory and fluctuation scales – showed relatively few differences over time, although as anticipated dementia with Lewy bodies groups had a greater degree of behavioural disturbance, more fluctuations and more parkinsonian symptoms than did individuals with non-Lewy body dementia.
Table 1 Clinical characteristics of participants at baseline and follow-up according to diagnosisa
| Probable DLB | Possible DLB | Non-DLB | No diagnosis | |
|---|---|---|---|---|
| Mini-Mental State Examination | ||||
| Baseline, mean (s.d.) | 20.9 (4.3) | 20.8 (4.4) | 21.7 (4.3) | 22.5 (2.9) |
| Follow-up, mean (s.d.) | 17.4 (6.5) | 19.0 (7.1) | 19.4 (6.1) | 19.8 (6.4) |
| Cambridge Cognitive Examination—revised version | ||||
| Baseline, mean (s.d.) | 63.0 (17.0) | 63.3 (16.6) | 66.4 (15.5) | 69.1 (12.7) |
| Follow-up, mean (s.d.) | 53.9 (20.5) | 55.9 (23.4) | 60.1 (20.4) | 64.1 (17.9) |
| Unified Parkinson's Disease Rating Scale | ||||
| Baseline, mean (s.d.) | 28.3 (14.5) | 17.4 (11.1) | 5.7 (7.7) | 16.6 (14.2) |
| Follow-up, mean (s.d.) | 36.0 (16.4) | 18.7 (11.6) | 6.5 (6.6) | 20.4 (22.2) |
| Hoehn and Yahr staging | ||||
| Baseline, mean (s.d.) | 2.44 (0.98) | 1.57 (1.02) | 0.22 (0.71) | 1.12 (1.36) |
| Follow-up, mean (s.d.) | 2.96 (1.07) | 1.69 (1.14) | 0.27 (0.81) | 1.43 (1.51) |
| Neuropsychiatric Inventory | ||||
| Baseline, mean (s.d.) | 22.1 (16.2) | 15.7 (11.2) | 10.1 (11.0) | 12.5 (10.8) |
| Follow-up, mean (s.d.) | 22.1 (17.7) | 17.8 (13.3) | 12.3 (12.8) | 10.7 (6.5) |
| Clinician Assessment of Fluctuation | ||||
| Baseline, mean (s.d.) | 6.6 (4.2) | 3.0 (4.3) | 0.3 (1.2) | 2.6 (4.1) |
| Follow-up, mean (s.d.) | 6.1 (4.2) | 1.8 (3.0) | 0.4 (1.7) | 1.0 (2.2) |
| Visual hallucinations | ||||
| Baseline, n (%)b | 55 (64) | 11 (44) | 5 (4) | N/A |
| Follow-up, n (%) | 58 (67) | 9 (36) | 2 (1.6) | N/A |
| Psycholepticsc | ||||
| Baseline, n (%) | 31 (33) | 20 (35) | 20 (14) | 10 (36) |
| Follow-up, n (%) | 35 (39) | 12 (44) | 22 (18) | 9 (43) |
| Psychoanalepticsd | ||||
| Baseline, n (%) | 69 (73) | 44 (77) | 106 (72) | 20 (71) |
| Follow-up, n (%) | 75 (84) | 17 (63) | 107 (85) | 21 (100) |
There were no significant differences noted for participants with and without follow-up data with respect to MMSE (mean=21.2 (s.d.=4.3) v. 19.2 (s.d.=4.3), P=0.99), UPDRS (mean=14.0 (s.d.=14.3) v. 20.5 (s.d.=14.7), P=0.76), CAMCOG–R (mean=64.6 (s.d.=16.2) v. 57.2 (s.d.=17.6), P=0.41), CAF (mean=2.8 (s.d.=4.2) v. 3.4 (s.d.=4.2); P=0.83) and severity of dementia (mean=1.22 (s.d.=0.67) v. 1.48 (s.d.=0.69), P=0.81).
Table 2 shows the comparison between consensus panel diagnosis at follow-up and baseline. Of the 68 individuals with probable dementia with Lewy bodies diagnosed at baseline, most retained their diagnosis at follow-up. In only five people (7%) was the diagnosis revised to possible dementia with Lewy bodies. Similarly, the diagnosis of non-Lewy body dementia showed relative stability. Possible dementia with Lewy bodies was, as anticipated, a more unstable diagnosis. Of the 44 individuals in this group, only 18 (41%) still had a diagnosis of possible dementia with Lewy bodies 1 year later, 26 had a clearer diagnosis at follow-up, 19 of probable dementia with Lewy bodies (43%) and 7 (16%) with non-Lewy body dementia.
Table 2 Changes in diagnosis over time

| Clinical diagnosis main assessment | Clinical diagnosis at follow-up (n=235), n (%) | |||
|---|---|---|---|---|
| n | Probable DLB | Possible DLB | Non-DLB | |
| Probable DLB | 68 | 63 (93) | 5 (7) | 0 |
| Possible DLB | 44 | 19 (43) | 18 (41) | 7 (16) |
| Non-DLB | 116 | 2 (2) | 1 (1) | 113 (97) |
| Other | 5 | 1 (20) | 1 (20) | 3 (60) |
| No diagnosis | 2 | 1 (50) | 0 | 1 (50) |
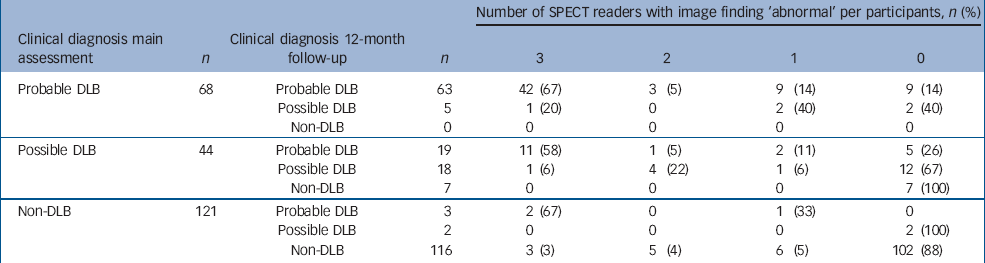
Table 3 shows results of 123I-FP-CIT SPECT ratings against final clinical diagnosis. Sensitivity ranged between 73.8 and 81.3% (mean=78) and specificity between 91.8 and 95.0% (mean=93.1). These findings are similar to those reported in the initial study (sensitivity mean, 77.7%; specificity, 90.4%). Reference McKeith, O'Brien, Walker, Tatsch, Booij and Darcourt5 The results of the scan ratings and final panel diagnoses are shown in Table 4. Of note is the possible dementia with Lewy bodies group, which was not included in the primary analysis of the baseline data. Nineteen participants diagnosed with possible dementia with Lewy bodies at baseline had their diagnosis revised to probable dementia with Lewy bodies at follow-up. Of these, 12 had an abnormal scan (as rated by at least two of the raters). By contrast, seven individuals were diagnosed with non-Lewy body dementia at follow-up, and all had normal scans. Although numbers are small, it is striking that 2 of the 3 individuals diagnosed with non-Lewy body dementia initially but who at follow-up were diagnosed with probable dementia with Lewy bodies had abnormal scans v. only 3 of 116 whose diagnosis of non-Lewy body dementia remained at follow-up. Similarly, only 1 of the 5 (20%) individuals in the probable dementia with Lewy bodies group who became a possible at follow-up had an abnormal scan, compared with 42 of 63 (67%) participants who remained in the probable dementia with Lewy bodies group. An abnormal scan, therefore, was predictive of an individual either keeping a diagnosis of dementia with Lewy bodies or the diagnosis becoming probable dementia with Lewy bodies.
Table 3 Sensitivity and specificity of 123I-FP-CIT SPECT v. follow-up clinical diagnosis

| Parameter | Reader | Participants with rateable scans, n | Participants with abnormal scans, n | Accuracy, % (95% CI) |
|---|---|---|---|---|
| Sensitivity | A | 80 | 65 | 81.3 (71.0-89.1) |
| B | 80 | 59 | 73.8 (62.7-83.0) | |
| C | 80 | 63 | 78.8 (68.2-87.1) | |
| Specificity | A | 121 | 115 | 95.0 (89.5-98.2) |
| B | 122 | 112 | 91.8 (85.4-96.0) | |
| C | 120 | 111 | 92.5 (86.2-96.5) |
Table 4 Changes in consensus panel diagnoses over time with the corresponding 123I-FP-CIT SPECT findings
| Number of SPECT readers with image finding ‘abnormal’ per participants, n (%) | |||||||
|---|---|---|---|---|---|---|---|
| Clinical diagnosis main assessment | n | Clinical diagnosis 12-month follow-up | n | 3 | 2 | 1 | 0 |
| Probable DLB | 68 | Probable DLB | 63 | 42 (67) | 3 (5) | 9 (14) | 9 (14) |
| Possible DLB | 5 | 1 (20) | 0 | 2 (40) | 2 (40) | ||
| Non-DLB | 0 | 0 | 0 | 0 | 0 | ||
| Possible DLB | 44 | Probable DLB | 19 | 11 (58) | 1 (5) | 2 (11) | 5 (26) |
| Possible DLB | 18 | 1 (6) | 4 (22) | 1 (6) | 12 (67) | ||
| Non-DLB | 7 | 0 | 0 | 0 | 7 (100) | ||
| Non-DLB | 121 | Probable DLB | 3 | 2 (67) | 0 | 1 (33) | 0 |
| Possible DLB | 2 | 0 | 0 | 0 | 2 (100) | ||
| Non-DLB | 116 | 3 (3) | 5 (4) | 6 (5) | 102 (88) | ||
Discussion
Differential diagnosis of Lewy body from non-Lewy body dementia
Our results, using follow-up diagnosis, confirm the high diagnostic accuracy of 123I-FP-CIT SPECT imaging in discriminating Lewy body from non-Lewy body dementia we previously reported using the baseline diagnosis. The 12-month follow-up was a powerful way of validating diagnosis in the uncertain group of people with possible dementia with Lewy bodies, since diagnosis became clearer in almost 60% of participants. At baseline, individuals with possible dementia with Lewy bodies had a bimodal distribution of 123I-FP-CIT uptake, Reference McKeith, O'Brien, Walker, Tatsch, Booij and Darcourt5 suggesting that the diagnosis reflected a heterogeneous population. The consensus panel's diagnoses at follow-up were not influenced by 123I-FP-CIT imaging results, as they relied solely on rating scores and information about symptoms from the site investigators. Strikingly, of the seven individuals with possible dementia with Lewy bodies deemed to be non-Lewy body dementia (all Alzheimer's disease) at follow-up, none had had an abnormal scan. By contrast, of the 19 people diagnosed with probable dementia with Lewy bodies at follow-up that had been categorised as possible dementia with Lewy bodies at baseline, 12 (63%) had abnormal 123I-FP-CIT scans. These results (63% sensitivity, 100% specificity), albeit in a small sample, mirror the overall results obtained for probable dementia with Lewy bodies. 123I-FP-CIT imaging, therefore, appears to have similar accuracy in discriminating Lewy body from non-Lewy body dementia in individuals with a possible diagnosis as it does in those with a probable diagnosis, and is likely to offer greatest diagnostic value when clinicians are most in need of support – namely, for accurate and timely diagnosis in clinically uncertain cases of individuals who have just one of the three core dementia with Lewy bodies features. These findings also support recent NICE guidelines for dementia, which suggest that 123I-FP-CIT SPECT imaging may be particularly helpful when the diagnosis of dementia with Lewy bodies is in doubt.
Stability of disease over time
The stability of the diagnosis of probable Alzheimer's disease is well recognised. However, far less attention has been given to longitudinal stability of a diagnosis of dementia with Lewy bodies. Our results show that, over 1 year, probable dementia with Lewy bodies is a stable diagnosis, as would be expected for a diagnosis with high specificity and positive predictive value as judged by clinicopathological studies. Reference Litvan, Bhatia, Burn, Goetz, Lang and McKeith2,Reference McKeith, Ballard, Perry, Ince, O'Brien and Neill21 The outcome of possible dementia with Lewy bodies has not previously been reported. In our study, only 40% of individuals with possible dementia with Lewy bodies at baseline remained in the possible category 12 months later. Our study did not include further assessments, so we cannot determine the proportion of participants whose diagnosis might be clear at 2 years. However, it is noteworthy (Table 4) that the proportion of abnormal 123I-FP-CIT scans in the possible dementia with Lewy bodies group had fallen in the year between baseline and follow-up. This finding is consistent with the view that most individuals diagnosed with possible dementia with Lewy bodies who did actually have dementia with Lewy bodies became apparent within 1 year.
Strengths and limitations
Strengths of this study include rigorous and quality-assured assessment of comprehensive clinical, cognitive and psychiatric ratings at baseline and follow-up, inclusion of multiple European sites, and use of a masked independent consensus panel for clinical diagnosis and independent masked readers for 123I-FP-CIT rating. A limitation is that, as with baseline findings, the gold standard for image validation was a clinical rather than a pathological diagnosis. However, the validity of the probable dementia with Lewy bodies diagnosis has been established in clinicopathological series, Reference Litvan, Bhatia, Burn, Goetz, Lang and McKeith2 and also in terms of 123I-FP-CIT changes both in our baseline and in other independent studies. Reference McKeith, O'Brien, Walker, Tatsch, Booij and Darcourt5–Reference Walker, Costa, Walker, Shaw, Gacinovic and Stevens7
Clinical implications
Assuming that our results can be extrapolated to the wider population of individuals with possible dementia with Lewy bodies, an abnormal 123I-FP-CIT scan in a person who fulfils possible dementia with Lewy bodies criteria strongly suggests a diagnosis of probable dementia with Lewy bodies and that the person should be managed as having dementia with Lewy bodies. The converse does not necessarily apply, since around 20% of individuals with probable dementia with Lewy bodies appear to have normal 123I-FP-CIT scans. There are a number of possible explanations for this finding. The clinical diagnosis may be inaccurate, some people with dementia with Lewy bodies may have normal dopaminergic function or the technique may be unable to detect subtle transporter loss (which might be more readily detected with partial volume effect correction). Reference Soret, Koulibaly, Darcourt and Buvat22 Further research needs to determine the proportion, clinical correlates and outcome in people with probable dementia with Lewy bodies who have normal 123I-FP-CIT imaging. This will require studies with repeat imaging (to determine at what point in the illness 123I-FP-CIT abnormalities may become apparent) as well as more studies involving autopsy verification. Of note, in the expanded series reported by Walker et al, Reference Walker, Jaros, Walker, Lee, Costa and Livingston23 123I-FP-CIT imaging was in agreement with autopsy diagnosis in the vast majority of cases (19/20), and was more precise than clinical diagnosis. This finding suggests that 123I-FP-CIT imaging may be more accurate than clinical diagnosis in distinguishing Lewy body from non-Lewy body dementia.
The latest revision of the International Consensus Criteria for dementia with Lewy bodies diagnosis incorporates abnormal 123I-FP-CIT imaging as a feature that, in addition to a single core feature, allows a diagnosis of probable dementia with Lewy bodies to be made. Reference McKeith, Dickson, Lowe, Emre, O'Brien and Feldman1 Results reported here confirm this is an appropriate and sensible amendment. Other investigations that may potentially assist with distinguishing dementia with Lewy bodies from Alzheimer's disease include 18F-flurodopa positron emission tomography (PET), which assesses the integrity of the nigrostriatal pathway, Reference Gilman, Koeppe, Little, An, Junck and Giordani24,Reference Hu, Okamura, Arai, Higuchi, Matsui and Tashiro25 18F-FDG PET, used to assess metabolic deficits, and abnormal MIBG (meta-iodobenzylguanidine) imaging, which assesses sympathetic cardiac denervation. Reference Hanyu, Shimizu, Hirao, Kanetaka, Iwamoto and Chikamori26,Reference Yoshita, Taki and Yamada27 Other differences of less certain diagnostic value include changes in cerebrospinal fluid measures of amyloid and tau, Reference Hampel, Buerger, Zinkowski, Teipel, Goernitz and Andreasen28 occipital hypoperfusion on SPECT or PET Reference Lobotesis, Fenwick, Phipps, Ryman, Swann and Ballard29 and preservation of medial temporal lobe structures on structural imaging. Reference Barber, Gholkar, Scheltens, Ballard, McKeith and O'Brien30 It may be that particular diffusion Reference Firbank, Blamire, Krishnan, Teodorczuk, English and Gholkar31 and spectroscopic Reference Kantarci, Petersen, Boeve, Knopman, Tang-Wai and O'Brien32 changes associated with dementia with Lewy bodies will emerge that can help distinguish it from Alzheimer's disease. However, currently 123I-FP-CIT SPECT imaging has the greatest evidence base. Several single-site studies and a large multicentre study have all demonstrated its utility for dementia with Lewy bodies diagnosis. Reference McKeith, O'Brien, Walker, Tatsch, Booij and Darcourt5 Our results confirm this position and demonstrate the diagnostic value of 123I-FP-CIT in clinically uncertain (possible) cases.
Acknowledgements
The sponsor (GE Healthcare) financially supported the collection, analysis and interpretation of data. The study was initiated by the principal investigators who drafted and approved the manuscript. The first author had full access to all the data in the study and wrote and approved the manuscript.










eLetters
No eLetters have been published for this article.