Tourette syndrome is a childhood-onset neuropsychiatric disorder defined by motor and vocal tics that persist for >1 year. In addition to having tics, children with Tourette syndrome often function poorly across numerous psychosocial domains Reference Carter, O'Donnell, Schultz, Scahill, Leckman and Pauls1 and have high rates of psychiatric comorbidity. Reference Kurlan, Como, Miller, Palumbo, Deeley and Andresen2 The long-term course of Tourette syndrome is variable, but tics typically improve during adolescence such that by age 18 years, up to 90% of individuals with Tourette syndrome have only mild tics or none at all. Reference Leckman, Zhang, Vitale, Lahnin, Lynch and Bondi3,Reference Pappert, Goetz, Louis, Blasucci and Leurgans4 It is unclear, however, whether their psychosocial functioning, like their tics, improves by this age. Previous longitudinal studies found that individuals with Tourette syndrome who were followed up in late adolescence or early adulthood functioned reasonably well, but these studies were limited by the absence of a control group. Reference Leckman, Bloch, King and Scahill5–Reference Burd, Kerbeshian, Barth, Klug, Avery and Benz7 In contrast, controlled cross-sectional studies reported that adults with Tourette syndrome had poorer quality of life Reference Elstner, Selai, Trimble and Robertson8 and more psychopathology, Reference Robertson, Banerjee, Hiley and Tannock9 but a critical limitation was that the participants were not necessarily identified as having Tourette syndrome in childhood and were attending specialty clinics as adults. Consequently, the poor outcomes described may apply mainly to the minority of individuals with Tourette syndrome who continue to have prominent tics and pursue treatment for them in adulthood. Our objective, therefore, was to reassess in late adolescence a large sample of individuals who were identified as having Tourette syndrome in childhood and may or may not still be in treatment, and compare them with community controls with respect to psychosocial functioning and psychiatric illness. We selected a community control group because we wanted to address a question that is salient for parents of children who present clinically with Tourette syndrome: how will my child function compared with individuals in my community who do not have Tourette syndrome? Our a priori hypothesis was that individuals with Tourette syndrome in late adolescence would have lower scores on the Children's Global Assessment Scale (CGAS). Reference Shaffer, Gould, Brasic, Ambrosini, Fisher and Bird10 We also explored whether psychosocial outcomes and rates of psychiatric comorbidity were associated with the severity of tic, attention-deficit hyperactivity disorder (ADHD) or obsessive–compulsive disorder (OCD) symptoms.
Method
Participants
Individuals with Tourette syndrome were recruited from 1993 to 1999 and consisted of two birth cohorts identified from all patients born in 1975 and 1980 and seen during childhood in the Yale Child Study Center Tourette syndrome and obsessive–compulsive disorder (OCD) clinic. To enable comparison between study participants and non-participants, families who did not participate in the in-person interviews could still complete the briefer telephone interview. Community controls were recruited from a list of 10 000 names purchased from a telemarketing company, and selected for contact using a random number generator. Controls were individually matched with those in the case group on age, gender, geographic region (ZIP code), and twin status (singleton v. twin). At the time of recruitment, participants were approximately 18 years old.
No exclusion criteria were stipulated for individuals with Tourette syndrome. Potential controls were excluded only if they reported a history of tics, or obsessive–compulsive symptoms or tics were observed on interview. After a complete description of the study, written informed consent was obtained from adult probands and parents, and assent was obtained from probands <18 years of age. Families were paid for participation. The institutional review boards at Yale University School of Medicine, New York State Psychiatric Institute, Columbia University College of Physicians and Surgeons and The Hospital for Sick Children approved the study.
Psychosocial outcome measures
Our primary psychosocial outcome measure was the CGAS, an adaptation of the Global Assessment Scale (GAS) Reference Endicott, Spitzer, Fleiss and Cohen11 for youth aged 4–16 years. It is a valid and reliable instrument used widely in research and clinically to assess overall psychiatric and social functioning of youth. Reference Shaffer, Gould, Brasic, Ambrosini, Fisher and Bird10,Reference Green, Shirk, Hanze and Wanstrath12 Children's Global Assessment Scale ratings range from 1 (lowest level of functioning) to 100 (highest). Although the mean (s.d.) age of our Tourette syndrome sample at reassessment was 18.1 (1.4) years (range 15.7–20.8), we selected the CGAS instead of a comparable scale for adults because we thought it would be superior for assessing older adolescents. Moreover, the theoretical reason for choosing 16 years as the upper age limit for the CGAS is not documented, and it has been used up to age 23 without reducing interrater reliability. Reference Schorre and Vandvik13
A consensus procedure was used by two child psychiatrists (D.A.G. and K.J.P.) to determine a CGAS score for each participant. They were masked to group status so that their ratings would not be influenced by knowledge of whether the individual had Tourette syndrome. Because a history of tics or obsessive–compulsive symptoms was the only exclusion criterion for potential controls, the raters had access to all collected information that did not refer to these symptoms. They determined CGAS scores independently and then conferred to resolve discrepancies ≥10 (the mean was used for discrepancies <10). The interrater reliability was high (intraclass correlation 0.8).
Secondary outcome measures included the Socialization domain of the Vineland Adaptive Behaviour Scales, Reference Sparrow, Balla and Cicchetti14 the Child Behaviour Checklist (CBCL) Reference Achenbach15 and the Youth Self-Report (YSR). Reference Achenbach16 The Vineland is a reliable and valid measure of personal and social functioning in youth aged 6–19 years. It is administered as a semi-structured interview with a primary caregiver. The CBCL is a reliable and valid parent-report measure for assessing problem behaviour and adaptive functioning in youth aged 4–18 years. The YSR, which also has adequate reliability and validity, is closely related to the CBCL and is designed to be completed by youth aged 11–18 years.
Assessment of psychiatric comorbidity
Another consensus procedure was used by two child psychiatrists (J.F.L. and B.S.P.) to determine lifetime DSM–IV 17 diagnoses for all participants. Again, the child psychiatrists were masked to group status and made determinations based on all data that did not refer to tics or obsessive–compulsive symptoms. After making diagnoses independently, they conferred to resolve differences. They also used the same procedure to confirm the diagnosis of Tourette syndrome (ignoring the impairment criterion) Reference Freeman, Fast, Burd, Kerbeshian, Robertson and Sandor18 in the individuals with Tourette syndrome.
Procedures for data collection
Demographic and clinical information was collected from the clinic chart and several other sources. The telephone interview, administered to a parent, included questions regarding the potential proband's history of tics, ADHD and obsessive–compulsive symptoms and learning. For individuals with potential Tourette syndrome, it also assessed current tic severity using the Yale Global Tic Severity Scale (YGTSS). Reference Leckman, Riddle, Hardin, Ort, Swartz and Stevenson19 The interviewer could not be masked to the group status of the potential proband because of the need to rule out a history of tics and obsessive–compulsive symptoms in potential controls, and to assess current tic severity in those with potential Tourette syndrome.
Parent and proband in-person interviews included the Revised Schedule for Tourette and Other Behavioural Syndromes (STOBS–R), a semi-structured interview used extensively in family-genetic studies. Reference Pauls and Hurst20 Parents were interviewed twice by different interviewers to prevent the assessment of tic and obsessive–compulsive symptoms during one interview from biasing the masked assessment of other symptoms and psychosocial functioning during the other. The former interview was not masked to group status and included sections of the Adult-on-Child STOBS-R pertaining to tic and obsessive–compulsive symptoms. For participants with Tourette syndrome, it also included assessments of current and worst-ever tic and OCD severity using the YGTSS and the Yale–Brown Obsessive Compulsive Scale (Y–BOCS). Reference Goodman, Price, Rasmusson, Mazure, Delgado and Henninger21,Reference Goodman, Price, Rasmusson, Mazure, Delgado and Henninger22 The other interview, where a different interviewer was masked to group status, included the Vineland Socialization domain and the portion of the Adult-on-Child STOBS–R that contains the Schedule for Affective Disorders and Schizophrenia for School-Age Children, Epidemiologic Version (Kiddie–SADS–E). Reference Orvaschel and Puig-Antich23 In the proband interview, the Child-on-Child STOBS–R was supplemented for individuals with Tourette syndrome with the YGTSS. The interviewer could not be masked to group status for any portion of the proband interview, because a proband's tics could be observed.
In addition to the CBCL (completed by parents) and YSR (completed by the proband), self-report measures included the ADHD Rating Scale–IV–Home Version Reference DuPaul, Power, Anastopoulos and Reid24 (completed separately by parents and the proband for current and worst-ever symptoms).
Despite our best efforts to collect all data for each participant, some families did not complete every secondary psychosocial outcome or symptom severity measure (completion rates are provided in online Table DS1).
Data analysis
Statistical analyses were performed using SPSS v.16.0 for Windows. In our primary analysis, CGAS scores were compared in the participants with Tourette syndrome and controls using analysis of covariance (ANCOVA). The groups were matched perfectly on gender, and we controlled statistically for age and socioeconomic status (quantified using the Hollingshead Four-Factor Index of Social Status). Reference Hollingshead25
All other analyses were considered exploratory. The approach used for the primary analysis was also used to assess group differences in the secondary psychosocial outcome measures (Vineland Socialization domain, CBCL and YSR). Group differences in lifetime rates of psychiatric disorders were assessed with chi-squared analyses and conditional logistic regression, applying the Cox regression option in SPSS and controlling for age and socioeconomic status. Because differences in outcome between groups might be related to the influence of ADHD, Reference Sukhodolsky, Scahill, Zhang, Peterson, King and Lombroso26 ANCOVA and conditional logistic regression analyses were repeated with the presence or severity of ADHD entered into the models as covariates. We also assessed the influence of ADHD by using ANCOVA to compare psychosocial outcomes in individuals with Tourette syndrome with and without ADHD and the controls (controlling for age, gender and socioeconomic status).
Within the Tourette syndrome group, ANCOVA was used to assess whether psychosocial outcomes were associated with a lifetime diagnosis of ADHD or OCD, and linear regression was used to assess whether these outcomes were associated with the severity of tic (based on the YGTSS excluding the impairment scale), ADHD or obsessive–compulsive symptoms. Similarly, logistic regression was used to assess whether comorbid psychiatric disorders were associated with the same variables. Age, gender and socioeconomic status were entered into the models as covariates. The significance level for all analyses was P = 0.05 (two-tailed). This was not adjusted for multiple comparisons because we had only one a priori hypothesis and the rest of the analyses were exploratory.
Results
Group characteristics
The sample comprised 65 individuals with Tourette syndrome and 65 community controls. Few potential controls were excluded on account of tics or obsessive–compulsive symptoms being discovered. Participants with Tourette syndrome received their initial clinical evaluation at a mean age of 10.8 (s.d. = 2.8) years and were reassessed 7.3 (s.d. = 3.1) years later. Non-participants with Tourette syndrome included 1 individual who died of a cerebral malignancy, 12 who refused to participate and 2 whose reason for non-participation is unclear. Of the 14 surviving non-participants, 9 completed the telephone interview. The 65 participants with Tourette syndrome and these 9 non-participants were comparable on demographic and clinical variables (data are provided in online Table DS2). The Tourette syndrome group and controls were similar in age (mean 18.1 (s.d. = 1.4) v. 18.4 (1.7) years, t (128) = 1.2, P = 0.2) and socioeconomic status (mean Hollingshead score 49.6 (s.d. = 10.8) v. 52.2 (8.0), t (128) = 1.5, P = 0.1). Individual matching of the Tourette syndrome group with controls was perfect on gender (57 males per group) and twin status (five twins, including two twin pairs, per group). In each group, all participants but one were White. All measures of tic and OCD severity showed that current symptoms were significantly improved compared with worst-ever symptoms in the Tourette syndrome group, and the same was true for ADHD symptoms in the Tourette syndrome group and controls (current and worst-ever symptom severity data are provided in online Table DS3).
Group comparisons
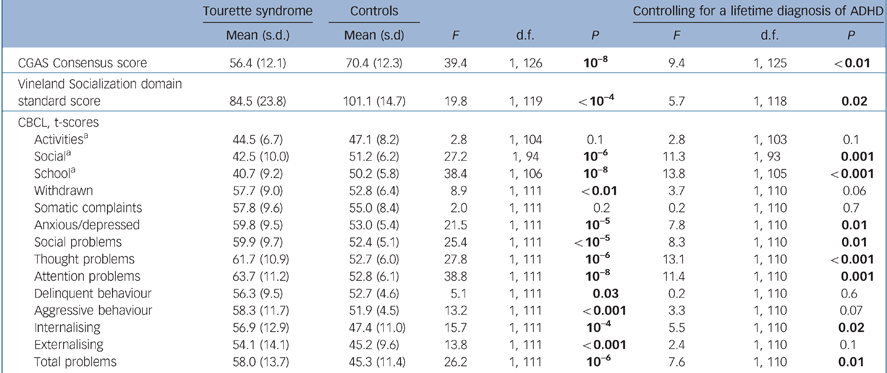
Scores on the CGAS, our primary psychosocial outcome measure, were significantly and substantially lower in the Tourette syndrome group than controls, and this difference remained significant when controlling for a lifetime diagnosis of ADHD (Table 1; complete data shown in online Table DS4) or any measure of ADHD severity (current or worst-ever symptoms rated by parents or probands). Similarly, Vineland Socialization standard scores were significantly lower in the Tourette syndrome group, and this difference also remained significant when controlling for each ADHD covariate. The CGAS scores correlated positively with Vineland Socialization scores (r = 0.5, P = 10–9).
Table 1 Comparison of Children's Global Assessment Scale (CGAS), Vineland Socialization domain and Child Behaviour Checklist (CBCL) scores in participants with Tourette syndrome and community controls
| Tourette syndrome | Controls | Controlling for a lifetime diagnosis of ADHD | ||||||
|---|---|---|---|---|---|---|---|---|
| Mean (s.d.) | Mean (s.d) | F | d.f. | P | F | d.f. | P | |
| CGAS Consensus score | 56.4 (12.1) | 70.4 (12.3) | 39.4 | 1, 126 | 10-8 | 9.4 | 1, 125 | < 0.01 |
| Vineland Socialization domain standard score | 84.5 (23.8) | 101.1 (14.7) | 19.8 | 1, 119 | < 10-4 | 5.7 | 1, 118 | 0.02 |
| CBCL, t-scores | ||||||||
| Activitiesa | 44.5 (6.7) | 47.1 (8.2) | 2.8 | 1, 104 | 0.1 | 2.8 | 1, 103 | 0.1 |
| Sociala | 42.5 (10.0) | 51.2 (6.2) | 27.2 | 1, 94 | 10-6 | 11.3 | 1, 93 | 0.001 |
| Schoola | 40.7 (9.2) | 50.2 (5.8) | 38.4 | 1, 106 | 10-8 | 13.8 | 1, 105 | < 0.001 |
| Withdrawn | 57.7 (9.0) | 52.8 (6.4) | 8.9 | 1, 111 | < 0.01 | 3.7 | 1, 110 | 0.06 |
| Somatic complaints | 57.8 (9.6) | 55.0 (8.4) | 2.0 | 1, 111 | 0.2 | 0.2 | 1, 110 | 0.7 |
| Anxious/depressed | 59.8 (9.5) | 53.0 (5.4) | 21.5 | 1, 111 | 10-5 | 7.8 | 1, 110 | 0.01 |
| Social problems | 59.9 (9.7) | 52.4 (5.1) | 25.4 | 1, 111 | < 10-5 | 8.3 | 1, 110 | 0.01 |
| Thought problems | 61.7 (10.9) | 52.7 (6.0) | 27.8 | 1, 111 | 10-6 | 13.1 | 1, 110 | < 0.001 |
| Attention problems | 63.7 (11.2) | 52.8 (6.1) | 38.8 | 1, 111 | 10-8 | 11.4 | 1, 110 | 0.001 |
| Delinquent behaviour | 56.3 (9.5) | 52.7 (4.6) | 5.1 | 1, 111 | 0.03 | 0.2 | 1, 110 | 0.6 |
| Aggressive behaviour | 58.3 (11.7) | 51.9 (4.5) | 13.2 | 1, 111 | < 0.001 | 3.3 | 1, 110 | 0.07 |
| Internalising | 56.9 (12.9) | 47.4 (11.0) | 15.7 | 1, 111 | 10-4 | 5.5 | 1, 110 | 0.02 |
| Externalising | 54.1 (14.1) | 45.2 (9.6) | 13.8 | 1, 111 | < 0.001 | 2.4 | 1, 110 | 0.1 |
| Total problems | 58.0 (13.7) | 45.3 (11.4) | 26.2 | 1, 111 | 10-6 | 7.6 | 1, 110 | 0.01 |
On the CBCL, participants with Tourette syndrome had lower t-scores on all competence subscales and higher t-scores on all syndrome subscales than controls, and most of these differences were highly significant. Moreover, internalising, externalising, and total problems t-scores were significantly higher in individuals with Tourette syndrome. When we controlled for ADHD diagnosis or severity, many of the group differences on the CBCL remained significant.
On the YSR, the Tourette syndrome group and controls had similar t-scores on all syndrome subscales, except for significantly higher attention problems in those with Tourette syndrome (mean = 58.3 (s.d. = 9.8) v. 54.8 (6.2), F = 4.6 (1, 103), P = 0.03). In contrast, those in the Tourette syndrome group had significantly lower t-scores on both competence subscales, activities (mean = 45.0 (s.d. = 7.8) v. 49.6 (6.3), F = 13.9 (1, 88), P<0.001) and social (mean = 46.8 (s.d. = 8.1) v. 51.1 (6.9), F = 6.6 (1, 84), P = 0.01). Differences on the attention problems subscale and the social competence subscale were no longer significant, however, when we controlled for ADHD diagnosis or parent-rated ADHD severity (current or worst-ever). No significant differences were found in internalising, externalising or total problems t-scores.
Compared with controls and participants with Tourette syndrome but not ADHD, those with Tourette syndrome and ADHD had significantly lower scores on the CGAS and Vineland Socialization domain, and significantly higher t-scores on the CBCL internalising, externalising, and total problems scales and the YSR externalising scale. In contrast, those in the Tourette syndrome group without ADHD and the controls did not differ significantly from each other on these measures, although they did differ significantly on several CBCL subscales (online Table DS5). These results suggest that the poorer psychosocial outcomes in the Tourette syndrome group were largely attributable to comorbid ADHD rather than Tourette syndrome itself; however, they are based on analyses that have lower statistical power than those presented in online Table DS4, which indicate that even after we controlled for the influence of ADHD, participants with Tourette syndrome still had impaired functioning compared with controls.
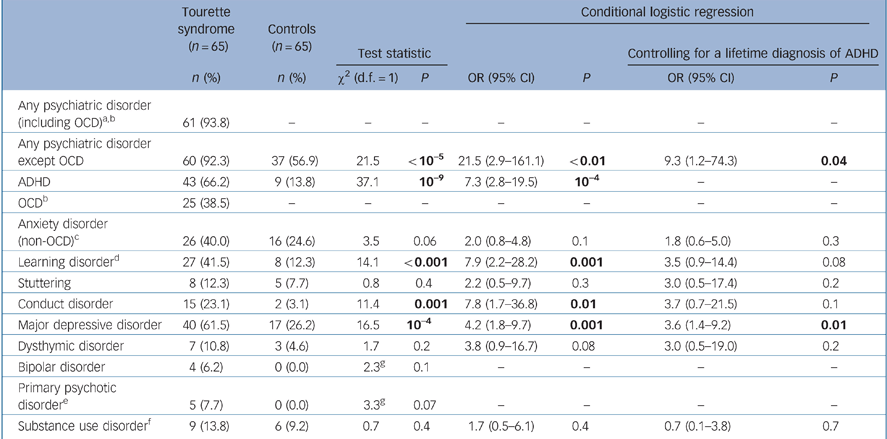
The Tourette syndrome group had significantly higher lifetime rates than controls of ADHD, learning disorder, conduct disorder and major depression, and tended to have higher rates of non-OCD anxiety disorders (Table 2). When we controlled, however, for a lifetime diagnosis of ADHD or parent-rated ADHD severity (current or worst-ever), group differences in rates of learning disorder and conduct disorder were no longer significant.
Table 2 Comparison of lifetime psychiatric disorders in the Tourette syndrome group and community controls
| Tourette syndrome (n = 65) | Controls (n = 65) | Conditional logistic regression | ||||||
|---|---|---|---|---|---|---|---|---|
| Test statistic | Controlling for a lifetime diagnosis of ADHD | |||||||
| n (%) | n (%) | χ2 (d.f. = 1) | P | OR (95% CI) | P | OR (95% CI) | P | |
| Any psychiatric disorder (including OCD)a,b | 61 (93.8) | — | — | — | — | — | — | — |
| Any psychiatric disorder except OCD | 60 (92.3) | 37 (56.9) | 21.5 | < 10-5 | 21.5 (2.9-161.1) | < 0.01 | 9.3 (1.2-74.3) | 0.04 |
| ADHD | 43 (66.2) | 9 (13.8) | 37.1 | 10-9 | 7.3 (2.8-19.5) | 10-4 | — | — |
| OCDb | 25 (38.5) | — | — | — | — | — | — | — |
| Anxiety disorder (non-OCD)c | 26 (40.0) | 16 (24.6) | 3.5 | 0.06 | 2.0 (0.8-4.8) | 0.1 | 1.8 (0.6-5.0) | 0.3 |
| Learning disorderd | 27 (41.5) | 8 (12.3) | 14.1 | < 0.001 | 7.9 (2.2-28.2) | 0.001 | 3.5 (0.9-14.4) | 0.08 |
| Stuttering | 8 (12.3) | 5 (7.7) | 0.8 | 0.4 | 2.2 (0.5-9.7) | 0.3 | 3.0 (0.5-17.4) | 0.2 |
| Conduct disorder | 15 (23.1) | 2 (3.1) | 11.4 | 0.001 | 7.8 (1.7-36.8) | 0.01 | 3.7 (0.7-21.5) | 0.1 |
| Major depressive disorder | 40 (61.5) | 17 (26.2) | 16.5 | 10-4 | 4.2 (1.8-9.7) | 0.001 | 3.6 (1.4-9.2) | 0.01 |
| Dysthymic disorder | 7 (10.8) | 3 (4.6) | 1.7 | 0.2 | 3.8 (0.9-16.7) | 0.08 | 3.0 (0.5-19.0) | 0.2 |
| Bipolar disorder | 4 (6.2) | 0 (0.0) | 2.3g | 0.1 | — | — | — | — |
| Primary psychotic disordere | 5 (7.7) | 0 (0.0) | 3.3g | 0.07 | — | — | — | — |
| Substance use disorderf | 9 (13.8) | 6 (9.2) | 0.7 | 0.4 | 1.7 (0.5-6.1) | 0.4 | 0.7 (0.1-3.8) | 0.7 |
Within-group analyses for the Tourette syndrome group
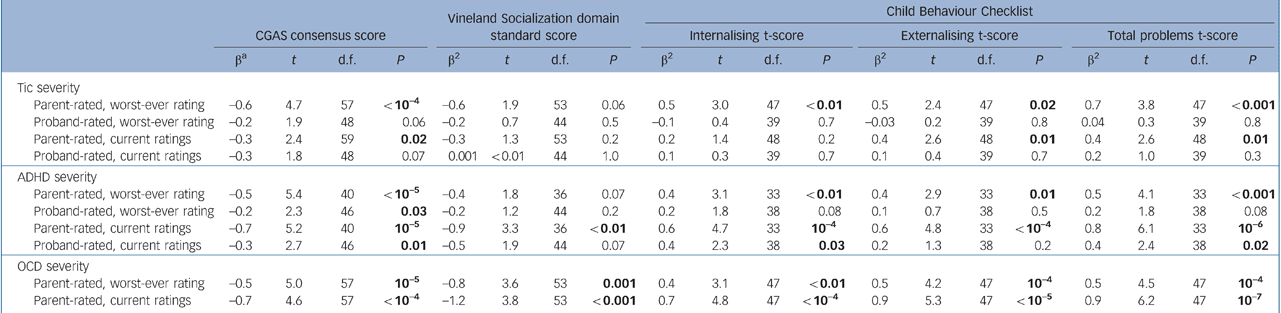
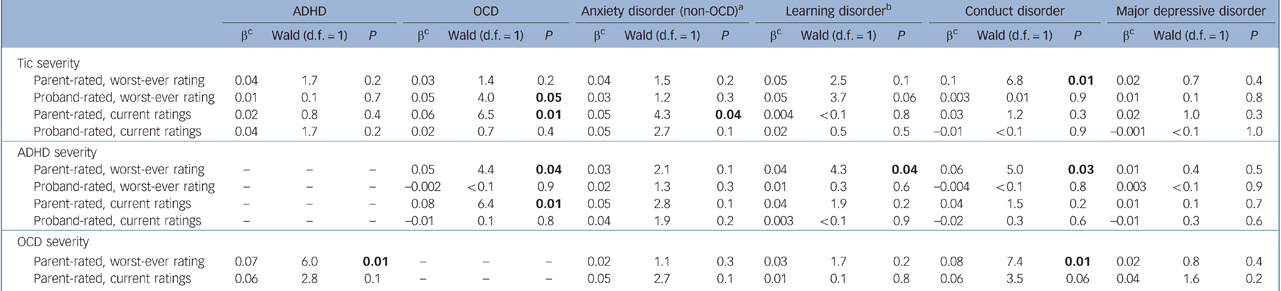
Within the Tourette syndrome group, associations of psychosocial outcomes with a lifetime diagnosis of ADHD and OCD and with tic, ADHD and obsessive–compulsive symptom severity are presented in Tables 3 and 4, and associations of comorbid psychiatric disorders with these variables are presented in Tables 5 and 6. Overall, worse psychosocial outcomes were associated with greater severity of tic, ADHD and obsessive–compulsive symptoms, particularly as rated by parents. Fewer associations were found between lifetime rates of psychiatric disorders and these measures. No symptom severity measure correlated with major depressive disorder.
Table 3 Participants with Tourette syndrome: associations of Children's Global Assessment Scale (CGAS), Vineland Socialization domain and Child Behaviour Checklist (CBCL) summary scale scores with lifetime diagnosis of attention-deficit hyperactivity disorder (ADHD) and obsessive–compulsive disorder (OCD)

| Child Behaviour Checklist | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CGAS consensus score | Vineland Socialization domain standard score | Internalising t-score | Externalising t-score | Total problems t-score | |||||||||||
| F | d.f. | P | F | d.f. | P | F | d.f. | P | F | d.f. | P | F | d.f. | P | |
| Lifetime diagnosis | |||||||||||||||
| ADHD | 27.7 | 1, 60 | < 10-5 | 5.5 | 1, 53 | 0.02 | 6.9 | 1, 48 | 0.01 | 9.4 | 1, 48 | < 0.01 | 13.5 | 1, 48 | 0.001 |
| OCD | 10.0 | 1, 60 | < 0.01 | 4.0 | 1, 53 | 0.05 | 1.0 | 1, 48 | 0.3 | 6.5 | 1, 48 | 0.01 | 4.0 | 1, 48 | 0.05 |
Table 4 Participants with Tourette syndrome: associations of Children's Global Assessment Scale (CGAS), Vineland Socialization domain and Child Behaviour Checklist (CBCL) summary scale scores with severity of tic, attention-deficit hyperactivity disorder (ADHD) and obsessive–compulsive disorder (OCD) symptoms
| Child Behaviour Checklist | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CGAS consensus score | Vineland Socialization domain standard score | Internalising t-score | Externalising t-score | Total problems t-score | ||||||||||||||||
| βa | t | d.f. | P | β2 | t | d.f. | P | β2 | t | d.f. | P | β2 | t | d.f. | P | β2 | t | d.f. | P | |
| Tic severity | ||||||||||||||||||||
| Parent-rated, worst-ever rating | -0.6 | 4.7 | 57 | < 10-4 | -0.6 | 1.9 | 53 | 0.06 | 0.5 | 3.0 | 47 | < 0.01 | 0.5 | 2.4 | 47 | 0.02 | 0.7 | 3.8 | 47 | < 0.001 |
| Proband-rated, worst-ever rating | -0.2 | 1.9 | 48 | 0.06 | -0.2 | 0.7 | 44 | 0.5 | -0.1 | 0.4 | 39 | 0.7 | -0.03 | 0.2 | 39 | 0.8 | 0.04 | 0.3 | 39 | 0.8 |
| Parent-rated, current ratings | -0.3 | 2.4 | 59 | 0.02 | -0.3 | 1.3 | 53 | 0.2 | 0.2 | 1.4 | 48 | 0.2 | 0.4 | 2.6 | 48 | 0.01 | 0.4 | 2.6 | 48 | 0.01 |
| Proband-rated, current ratings | -0.3 | 1.8 | 48 | 0.07 | 0.001 | < 0.01 | 44 | 1.0 | 0.1 | 0.3 | 39 | 0.7 | 0.1 | 0.4 | 39 | 0.7 | 0.2 | 1.0 | 39 | 0.3 |
| ADHD severity | ||||||||||||||||||||
| Parent-rated, worst-ever rating | -0.5 | 5.4 | 40 | < 10-5 | -0.4 | 1.8 | 36 | 0.07 | 0.4 | 3.1 | 33 | < 0.01 | 0.4 | 2.9 | 33 | 0.01 | 0.5 | 4.1 | 33 | < 0.001 |
| Proband-rated, worst-ever rating | -0.2 | 2.3 | 46 | 0.03 | -0.2 | 1.2 | 44 | 0.2 | 0.2 | 1.8 | 38 | 0.08 | 0.1 | 0.7 | 38 | 0.5 | 0.2 | 1.8 | 38 | 0.08 |
| Parent-rated, current ratings | -0.7 | 5.2 | 40 | 10-5 | -0.9 | 3.3 | 36 | < 0.01 | 0.6 | 4.7 | 33 | 10-4 | 0.6 | 4.8 | 33 | < 10-4 | 0.8 | 6.1 | 33 | 10-6 |
| Proband-rated, current ratings | -0.3 | 2.7 | 46 | 0.01 | -0.5 | 1.9 | 44 | 0.07 | 0.4 | 2.3 | 38 | 0.03 | 0.2 | 1.3 | 38 | 0.2 | 0.4 | 2.4 | 38 | 0.02 |
| OCD severity | ||||||||||||||||||||
| Parent-rated, worst-ever rating | -0.5 | 5.0 | 57 | 10-5 | -0.8 | 3.6 | 53 | 0.001 | 0.4 | 3.1 | 47 | < 0.01 | 0.5 | 4.2 | 47 | 10-4 | 0.5 | 4.5 | 47 | 10-4 |
| Parent-rated, current ratings | -0.7 | 4.6 | 57 | < 10-4 | -1.2 | 3.8 | 53 | < 0.001 | 0.7 | 4.8 | 47 | < 10-4 | 0.9 | 5.3 | 47 | < 10-5 | 0.9 | 6.2 | 47 | 10-7 |
Table 5 Participants with Tourette Syndrome: associations of selected lifetime psychiatric disorders with lifetime diagnosis of attention-deficit hyperactivity disorder (ADHD) and obsessive–compulsive disorder (OCD)

| ADHD | OCD | Anxiety disorder (non-OCD)a | Learning disorderb | Conduct disorder | Major depressive disorder | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P | |
| Lifetime diagnosis | ||||||||||||
| ADHD | - | - | 2.4 (0.7-7.8) | 0.2 | 3.4 (0.9-12.5) | 0.07 | 3.9 (1.1-14.1) | 0.04 | 2.4 (0.6-10.3) | 0.3 | 1.5 (0.5-4.7) | 0.5 |
| OCD | 2.4 (0.7-7.9) | 0.2 | - | - | 3.0 (1.0-9.2) | 0.05 | 2.3 (0.8-6.8) | 0.1 | 3.8 (1.1-13.5) | 0.04 | 2.1 (0.7-6.3) | 0.2 |
Table 6 Participants with Tourette Syndrome: associations of selected lifetime psychiatric disorders with severity of tic, attention-deficit hyperactivity disorder (ADHD) and obsessive–compulsive disorder (OCD) symptoms
| ADHD | OCD | Anxiety disorder (non-OCD)a | Learning disorderb | Conduct disorder | Major depressive disorder | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| βc | Wald (d.f. = 1) | P | βc | Wald (d.f. = 1) | P | βc | Wald (d.f. = 1) | P | βc | Wald (d.f. = 1) | P | βc | Wald (d.f. = 1) | P | βc | Wald (d.f. = 1) | P | |
| Tic severity | ||||||||||||||||||
| Parent-rated, worst-ever rating | 0.04 | 1.7 | 0.2 | 0.03 | 1.4 | 0.2 | 0.04 | 1.5 | 0.2 | 0.05 | 2.5 | 0.1 | 0.1 | 6.8 | 0.01 | 0.02 | 0.7 | 0.4 |
| Proband-rated, worst-ever rating | 0.01 | 0.1 | 0.7 | 0.05 | 4.0 | 0.05 | 0.03 | 1.2 | 0.3 | 0.05 | 3.7 | 0.06 | 0.003 | 0.01 | 0.9 | 0.01 | 0.1 | 0.8 |
| Parent-rated, current ratings | 0.02 | 0.8 | 0.4 | 0.06 | 6.5 | 0.01 | 0.05 | 4.3 | 0.04 | 0.004 | <0.1 | 0.8 | 0.03 | 1.2 | 0.3 | 0.02 | 1.0 | 0.3 |
| Proband-rated, current ratings | 0.04 | 1.7 | 0.2 | 0.02 | 0.7 | 0.4 | 0.05 | 2.7 | 0.1 | 0.02 | 0.5 | 0.5 | -0.01 | <0.1 | 0.9 | -0.001 | <0.1 | 1.0 |
| ADHD severity | ||||||||||||||||||
| Parent-rated, worst-ever rating | - | - | - | 0.05 | 4.4 | 0.04 | 0.03 | 2.1 | 0.1 | 0.04 | 4.3 | 0.04 | 0.06 | 5.0 | 0.03 | 0.01 | 0.4 | 0.5 |
| Proband-rated, worst-ever rating | - | - | - | -0.002 | <0.1 | 0.9 | 0.02 | 1.3 | 0.3 | 0.01 | 0.3 | 0.6 | -0.004 | <0.1 | 0.8 | 0.003 | <0.1 | 0.9 |
| Parent-rated, current ratings | - | - | - | 0.08 | 6.4 | 0.01 | 0.05 | 2.8 | 0.1 | 0.04 | 1.9 | 0.2 | 0.04 | 1.5 | 0.2 | 0.01 | 0.1 | 0.7 |
| Proband-rated, current ratings | - | - | - | -0.01 | 0.1 | 0.8 | 0.04 | 1.9 | 0.2 | 0.003 | <0.1 | 0.9 | -0.02 | 0.3 | 0.6 | -0.01 | 0.3 | 0.6 |
| OCD severity | ||||||||||||||||||
| Parent-rated, worst-ever rating | 0.07 | 6.0 | 0.01 | - | - | - | 0.02 | 1.1 | 0.3 | 0.03 | 1.7 | 0.2 | 0.08 | 7.4 | 0.01 | 0.02 | 0.8 | 0.4 |
| Parent-rated, current ratings | 0.06 | 2.8 | 0.1 | - | - | - | 0.05 | 2.7 | 0.1 | 0.01 | 0.1 | 0.8 | 0.06 | 3.5 | 0.06 | 0.04 | 1.6 | 0.2 |
Discussion
The major finding of this study, confirming our a priori hypothesis, is that the global functioning of older adolescents with Tourette syndrome who were identified in childhood was substantially poorer than that of community controls. Furthermore, their socialisation, according to the Vineland, was worse; they had more internalising and externalising problems according to the CBCL; and they had higher lifetime rates of other psychiatric disorders. Much of the impairment in individuals with Tourette syndrome, however, was attributable to comorbid ADHD.
Several features of the study strengthen the validity of these findings. First, the parent in-person interview was divided into two parts, with the interviewer masked to group status during the portion that included the Vineland and Kiddie–SADS–E. Second, our Tourette syndrome sample is representative of the target population, as we recruited >80% of eligible individuals to participate and determined that non-participants were similar to participants on demographic and clinical measures. Third is the narrow age range of participants, as studies of psychiatric comorbidity based on lifetime prevalence in mixed-age samples can create the appearance of comorbidity even when disorders are randomly associated. Reference Kraemer, Wilson and Hayward27 Finally, we compared participants with Tourette syndrome with community controls who were matched on demographic variables and ascertained with few exclusion criteria; therefore, differences in outcome are unlikely to be related to confounding factors or to selection of controls who are particularly high functioning. Indeed, the mean CGAS score in the control group was right at the cut-off point for normal function (>70) suggested by the instrument's authors. Reference Shaffer, Gould, Brasic, Ambrosini, Fisher and Bird10
Our finding of impaired global functioning in individuals with Tourette syndrome reassessed in late adolescence counters results of longitudinal studies that found relatively high functional status around this age. Reference Leckman, Bloch, King and Scahill5–Reference Burd, Kerbeshian, Barth, Klug, Avery and Benz7 Those studies were limited, however, by the absence of a control group, and rates of comorbid ADHD were much lower than in our sample. Our study also extends results of controlled cross-sectional studies in which children or adults with Tourette syndrome were found to have worse functioning than controls, particularly if they had comorbid ADHD, Reference Carter, O'Donnell, Schultz, Scahill, Leckman and Pauls1,Reference Elstner, Selai, Trimble and Robertson8,Reference Sukhodolsky, Scahill, Zhang, Peterson, King and Lombroso26 and higher rates of comorbid psychopathology. Reference Robertson, Banerjee, Hiley and Tannock9,Reference Khalifa and von Knorring28 Furthermore, whereas previous studies of adults with Tourette syndrome recruited individuals from patients attending a specialty clinic, Reference Elstner, Selai, Trimble and Robertson8,Reference Robertson, Banerjee, Hiley and Tannock9 the Tourette syndrome group in our study was identified in childhood and then contacted for reassessment years later. As a result, we can be confident that our Tourette syndrome group does not represent a subgroup of individuals with Tourette syndrome with particularly severe or persistent tics for which they were pursuing treatment. On the contrary, their tics at the time of reassessment were generally mild.
Associations of outcomes with tic severity
The relationship between tic severity and psychosocial functioning in people with Tourette syndrome remains unclear, despite multiple attempts to examine it. Many cross-sectional reports on youth indicate that those with more severe tics have worse functioning, Reference Coffey, Biederman, Geller, Spencer, Kim and Bellordre29,Reference Storch, Merlo, Lack, Milsom, Geffken and Goodman30 but many others indicate no association. Reference Carter, O'Donnell, Schultz, Scahill, Leckman and Pauls1,Reference Sukhodolsky, Scahill, Zhang, Peterson, King and Lombroso26,Reference Khalifa and von Knorring28,Reference Hoekstra, Steenhuis, Troost, Korf, Kallenberg and Minderaa31 The variability in findings is likely related to the wide age range of the samples, the complicated trajectory of tics during childhood, Reference Leckman, Zhang, Vitale, Lahnin, Lynch and Bondi3 the large fluctuation in tic severity at any given age, and variable statistical power to detect real effects. In keeping with limited longitudinal data, Reference Leckman, Bloch, King and Scahill5,Reference Bloch, Peterson, Scahill, Otka, Katsovich and Zhang6 however, we found that lower CGAS scores in late adolescence, determined by evaluators masked to tic history, were associated with greater tic severity as rated by parents. Similarly, higher CBCL internalising, externalising, and total problems scores were associated with more severe parent-rated tics. In contrast, we found few associations in older adolescents between lifetime rates of comorbid illnesses and measures of tic severity. A notable exception was that a lifetime diagnosis of OCD was associated with more severe parent-rated current tics, suggesting that children with Tourette syndrome and comorbid OCD may be more likely to have more severe tic symptoms in late adolescence.
Associations of outcomes with ADHD
With few exceptions, Reference Coffey, Biederman, Geller, Spencer, Kim and Bellordre29 studies of youth with Tourette syndrome indicate that comorbid ADHD is strongly associated with poorer psychosocial outcomes, Reference Carter, O'Donnell, Schultz, Scahill, Leckman and Pauls1,Reference Sukhodolsky, Scahill, Zhang, Peterson, King and Lombroso26,Reference Hoekstra, Steenhuis, Troost, Korf, Kallenberg and Minderaa31,Reference Spencer, Biederman, Harding, O'Donnell, Wilens and Faraone32 and our findings support this conclusion. Nevertheless, after we adjusted for the influence of ADHD, we still found that older adolescents with Tourette syndrome had impaired functioning compared with controls. These results are consistent with a recent cross-sectional study of adults with Tourette syndrome, which found that those with a history of ADHD had significantly more psychopathology and maladaptive behaviours, but those without ADHD still had higher levels of psychopathology compared with population norms. Reference Haddad, Umoh, Bhatia and Robertson33 Limited data regarding the association of ADHD with other comorbid disorders in individuals with Tourette syndrome have yielded mixed results, Reference Sukhodolsky, Scahill, Zhang, Peterson, King and Lombroso26,Reference Spencer, Biederman, Harding, O'Donnell, Wilens and Faraone32 but within-group analyses in our Tourette syndrome sample indicate that parent-rated worst-ever ADHD severity was associated with OCD, learning disorder and conduct disorder, although not major depressive disorder. Moreover, between-group analyses reveal that ADHD diagnosis and severity accounted for elevated rates in those with Tourette syndrome of learning disorder and conduct disorder, but again not major depressive disorder. We cannot conclude from our data whether an inherent, neurobiological relationship exists between ADHD and the other disorders associated with it, or whether the influence of ADHD on psychosocial functioning increases the risk that they will develop.
Associations of outcomes with OCD
Some Reference Carter, O'Donnell, Schultz, Scahill, Leckman and Pauls1,Reference Lin, Katsovich, Ghebremichael, Findley, Grantz and Lombroso34 but not all Reference Coffey, Biederman, Geller, Spencer, Kim and Bellordre29,Reference Hoekstra, Steenhuis, Troost, Korf, Kallenberg and Minderaa31 studies indicate that comorbid OCD is associated with poorer psychosocial functioning in youth with Tourette syndrome. The inconsistency likely stems from the young and variable ages of the samples in relation to the usual adolescent age at onset of OCD in the context of pre-existing Tourette syndrome. Reference Bloch, Peterson, Scahill, Otka, Katsovich and Zhang6 We found, however, that not only current but also worst-ever OCD severity was associated with poorer functioning in late adolescence according to the CGAS, Vineland and CBCL, suggesting that OCD in childhood can have long-term psychosocial repercussions. We also found that a history of OCD was associated with higher lifetime rates of conduct disorder and non-OCD anxiety disorders, and patients with more severe obsessive–compulsive symptoms were more likely to have conduct disorder and ADHD. Although a previous study of adults with Tourette syndrome found no meaningful differences between those with and without comorbid OCD in rates of other comorbid disorders, Reference Coffey, Miguel, Biederman, Baer, Rauch and O'Sullivan35 that study had a smaller sample, did not assess for conduct disorder and did not report associations with OCD severity. Moreover, our results are consistent with a previous report that OCD was associated with higher rates of other anxiety disorders in youth with Tourette syndrome, Reference Coffey, Biederman, Smoller, Geller, Sarin and Schwartz36 and with epidemiologically based findings that OCD was associated with prior separation anxiety disorder, ADHD and conduct problems. Reference Peterson, Pine, Cohen and Brook37
Relationship of Tourette syndrome with major depression
Our finding that nearly two-thirds of older adolescents with Tourette syndrome had a lifetime diagnosis of major depressive disorder is consistent with many previous reports indicating a strong association between Tourette syndrome and depression in clinical samples. Reference Robertson38 The reason for this association, however, remains a source of controversy. Some maintain that depression is an integral part of Tourette syndrome and shares a common genetic basis, Reference Comings and Comings39 whereas others argue that the disorders are genetically unrelated, and that Tourette syndrome is a risk factor for depression because of the emotional consequences of having a chronic illness. Reference Pauls, Leckman and Cohen40 In keeping with the latter hypothesis, the influence of tic severity, ADHD, OCD and psychosocial stress have been thought to account for the association of Tourette syndrome with depression. Reference Lin, Katsovich, Ghebremichael, Findley, Grantz and Lombroso34,Reference Robertson38 Our data, however, argue against this possibility, as within the Tourette syndrome group, major depressive disorder was the only comorbid disorder for which we found no association with a lifetime diagnosis of ADHD or OCD, or any measure of tic, ADHD or OCD severity. Moreover, unlike learning disorder and conduct disorder, the between-group difference in rates of major depressive disorder remained significant after controlling for ADHD diagnosis or severity. The association of Tourette syndrome with depression requires further investigation, but our data suggest that these disorders could be inherently linked, independent of the burden of chronic illness.
Limitations
As in most studies of Tourette syndrome, we recruited individuals from a specialty clinic, and therefore the psychosocial impairment and high rates of comorbidity we found may be partly related to Berkson's bias. Reference Berkson41 In addition, the findings may not be generalisable to all individuals with Tourette syndrome, especially those who do not present clinically and receive a diagnosis. Nevertheless, it is precisely the clinical nature of our sample that makes our findings clinically relevant, as they pertain to children whose parents are sufficiently concerned about their child's tic symptoms to pursue a clinical assessment. A sample of epidemiologically ascertained youth would include many whose symptoms were unrecognised or of little or no concern. Furthermore, although our Tourette syndrome group was recruited from a specialty clinic in a single tertiary care centre, most received their initial diagnostic assessment for Tourette syndrome at that same centre, and very few were referred to us because previously diagnosed Tourette syndrome was too challenging to be managed elsewhere. Consequently, our sample is likely similar to that of individuals who receive a diagnosis of Tourette syndrome in less specialised clinical settings. Most of our participants were middle-class and White, however, and therefore our findings may not generalise to other ethnic and socioeconomic groups. On the other hand, using a relatively privileged sample such as ours is likely to bias the results in favour of better outcomes, and even in our sample we found that older adolescents with Tourette syndrome experienced considerable morbidity.
The CGAS scores were determined retrospectively, and although the raters followed the instrument's guidelines, the exact scores assigned inevitably depended on the subjective judgement of the raters. Retrospective rating of the CGAS was necessary, however, to ensure that the raters were masked to group status. Furthermore, the CGAS's interrater and test–retest reliability, as well as its concurrent and discriminant validity, have been amply demonstrated. Reference Shaffer, Gould, Brasic, Ambrosini, Fisher and Bird10,Reference Green, Shirk, Hanze and Wanstrath12,Reference Schorre and Vandvik13 In our study, the interrater reliability of CGAS scores was likewise high, and their validity was supported by their significant correlation with Vineland Socialization scores. The assessments of worst-ever symptom severity were also retrospective and therefore could have been influenced by recall bias. Despite this possibility, it is reassuring that in the Tourette syndrome group the associations of psychosocial outcomes with parent-rated worst-ever ADHD and OCD severity were generally consistent with the associations of these outcomes with a lifetime diagnosis of ADHD and OCD (determined by two child psychiatrists and based on multiple sources of information).
The naturalistic design of our study did not permit an analysis of treatment effects, although 85% of participants with Tourette syndrome were treated pharmacologically at some time during the course of their illness. This treatment history is consistent with the psychosocial impairment that our participants experienced, and it suggests that the long-term benefits of pharmacotherapy may be limited for individuals with such complex psychopathology. Although we found that individuals with Tourette syndrome had impaired psychosocial functioning as older adolescents, assessment in later years is necessary to determine whether this impairment persists into adulthood.
In the interest of generating hypotheses and minimising the risk of Type II errors, we conducted multiple exploratory analyses without adjusting the significance level of P = 0.05. Therefore, we cannot rule out the possibility that some post hoc findings may have occurred by chance. However, even when we applied the more stringent significance level of P = 0.001 the overall pattern of results was largely unchanged.
Implications
Our study provides new information to help clinicians counsel parents whose children have Tourette syndrome. Previous reports indicate that children with Tourette syndrome generally have impaired psychosocial functioning that is attributable in large part to ADHD and possibly OCD and tic severity. Reference Carter, O'Donnell, Schultz, Scahill, Leckman and Pauls1,Reference Sukhodolsky, Scahill, Zhang, Peterson, King and Lombroso26,Reference Coffey, Biederman, Geller, Spencer, Kim and Bellordre29,Reference Storch, Merlo, Lack, Milsom, Geffken and Goodman30 The prognosis for psychosocial functioning, however, has been unclear. Our data show that older adolescents with Tourette syndrome who were identified in childhood continue to experience considerable psychosocial impairment, and they have high lifetime rates of ADHD, major depressive disorder, learning disorder, OCD, non-OCD anxiety disorders and conduct disorder. Thus, although parents can be reassured that tics are likely to improve by 18 years of age, Reference Leckman, Zhang, Vitale, Lahnin, Lynch and Bondi3,Reference Pappert, Goetz, Louis, Blasucci and Leurgans4 outcomes for psychosocial functioning and comorbid conditions are generally less optimistic. Our study also offers clinical clues to inform the prognosis for an individual child with Tourette syndrome. Poorer outcomes in late adolescence are associated not only with more severe ADHD symptoms in childhood, but also with more severe OCD symptoms and tics. In contrast, although the risk of developing major depressive disorder is high, it appears to be independent of the severity of these symptoms. Overall, our findings highlight the importance of closely monitoring children with Tourette syndrome as they mature for psychosocial impairment and comorbid conditions.
Funding
This work was supported in part by a grant from the March of Dimes (BSP); National Institutes of Health grant K02-74677 (BSP); and the Suzanne Crosby Murphy Endowment at Columbia University College of Physicians and Surgeons.
Acknowledgements
The authors thank Hongtu Zhu, PhD, for providing statistical consultation.











eLetters
No eLetters have been published for this article.